Ultrasound Imaging – Adenomyosis
Pierre Vassallo
INTRODUCTION
Adenomyosis is a common condition in which tissues of the endometrial lining (endometrial glands and stroma) migrate into the myometrium. Tese ectopic endometrial tissues incite uterine enlargement through myometrial hyperplasia and
hypertrophy. Tey also cause increased myometrial vascularity.
INCIDENCE
Adenomyosis has been reported to occur in around 20-30% of women.1 In the past, this disease was thought to occur mainly at around 40-50 years of age because it was most frequently diagnosed on hysterectomy specimens. However, with the advent of MRI and more recently, high resolution endovaginal ultrasound, this disease is now frequently diagnosed in 20-40-year-olds and in post-menopausal women.2
CAUSES
The cause for the migration of endometrial tissues into the myometrium is unknown. Proposed mechanisms include endometrial invagination, migration of endometrial tissues along myometrial lymphatics and embryologically displaced
Mullerian remnants. While there has been a clear association between adenomyosis and multiparity, patients with traits towards high oestrogen exposure such as early menarche and short menstrual cycles, and individuals with a high body-mass-index also show an increased risk.2
While the association with multiparity may be explained through high oestrogen exposure, migration of endometrial tissues may also result from trophoblastic penetration that occurs during placental growth.2
PATHOPHYSIOLOGY
The pathological fndings seen in adenomyosis can be categorised into three components: the “adeno” component involves migration of endometrial tissues into the myometrium, the “myosis” component is the reaction of the adjacent
myometrium that includes myometrial hyperplasia and hypertrophy, while the third component consists of increased myometrial vascularity.
SIGN AND SYMPTOMS
Approximately a third of adenomyosis cases are asymptomatic, being diagnosed only on endovaginal ultrasound. Te most common symptom is menorrhagia (heavy periods), which is thought to result because of the increased number of endometrial glands and increased myometrial vascularity. Te second most common symptom of adenomyosis is dysmenorrhoea (painful periods) that likely occurs because of intramyometrial bleeding and increased prostaglandin levels, which lead to myometrial contraction and vasoconstriction.
Other symptoms of adenomyosis include pelvic pain, metrorrhagia (irregular uterine bleeding most notably between menstrual periods) and dyspareunia (painful intercourse).
The association with infertility is thought to result from changes in the junctional zone of the endometrium that interfere with implantation. An increased rate of spontaneous abortion seen in patients with adenomyosis is likely due to increased
uterine contractions, endometrial inflammation and an altered hormonal environment.
Signs of adenomyosis are non-specifc including pelvic tenderness and uterine enlargement, which are also seen with uterine fbroids and endometriosis.
ULTRASOUND (US) FINDINGS
US fndings may be classifed based on the three pathological mechanisms described above.
A. Te US fndings related to migration of the endometrial glands include a loss of defnition of the endometrial/myometrial interface, a thickened inner myometrial layer, myometrial heterogeneity and myometrial cysts (Fig 1). Tese cysts are typically 1-5mm in diameter and are usually anechoic. However, since they contain endometrial glands, they may contain echogenic material representing blood degeneration products. Occasionally a cyst may connect to the endometrium through a long duct (“lollipop diverticulum”).
B. Since migrated endometrial glands are hormonally active, they induce a local reaction (myosis) in adjacent myometrium. This is seen as thickening of the inner myometrium on US, which may sometimes demonstrate myometrial striations
(Fig 2). Focal myometrial thickening (Fig 3) may resemble a fbroid, however the latter usually has more distinct margins. Diffuse uterine enlargement may also occur. The portion of the myometrium affected by adenomyosis shows a course
heterogeneous texture with thin vertical shadows that has been termed a venetian blind appearance (Fig 4).
C. Increased vascularity of the myometrium at the site of adenomyosis can be readily seen on colour Doppler US imaging (Fig 5). Te increased vascularity is reactive to muscle hyperplasia and hypertrophy. Vascularity tends to be central in adenomyosis and peripheral in a fbroid (Fig 6). The best way to visualise the above fndings are with high frequency endovaginal probes. However, with a large uterus, the settings may need to be adjusted to lower frequencies to penetrate
to the posterior myometrium. Occasionally, transabdominal pelvic US may be required for very large uteruses. Using 3D US to generate reconstructions of the uterus in any plane has also been suggested by some authors, with limited degrees of success.
Saline-infusion sono-hysterography (SIS) is a technique whereby saline is injected into the endometrial cavity during US examination to better assess the endometrial lining. It is the US equivalent of X-ray hysterosalpingography,
which avoids ionizing radiation. SIS may be beneficial in equivocal cases of adenomyosis as it clearly demonstrates the endometrium and may show filling of the endometrial glands with saline (Fig 7).
In short, adenomyosis is a common cause of dysmenorrhoea, menorrhagia and metrorrhagia. It may also cause infertility. High-frequency endovaginal ultrasound provides efficient confirmation of this condition and should be the frst test if the
condition is suspected. Tis imaging technique readily detects the disease and serves to distinguish other gynaecological causes of pelvic pain such as adnexal cysts, ectopic gestation and pelvic inflammatory disease.
References
1. Vercellini P, Viganò P, Somigliana E et al. Adenomyosis: epidemiological factors. Best Pract Res Clin Obstet Gynaecol 2006; 20(4):465-77.
2. Struble J, Reid S, Bedaiwy MA. Adenomyosis: a clinical review of a challenging gynecologic condition. J Minim Invasive Gynecol 2016; 23(2):164-85.
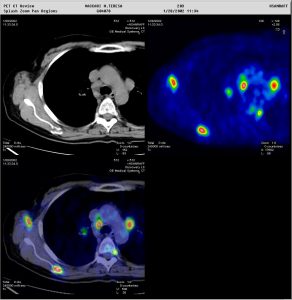
Figure 1: Staging and re-staging using 18F-FDG. Metabolic PET image superimposed on CT image helps the nuclear physician to report with confidence (Images courtesy of HSR, Milano)
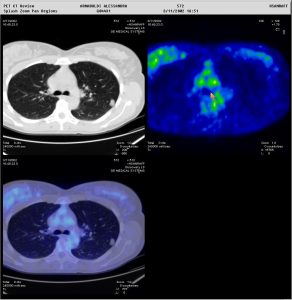
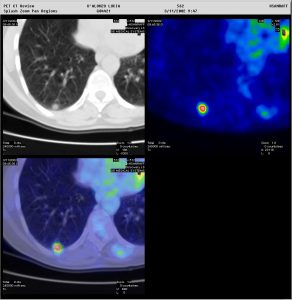
Figure 2: Two patients presenting with a pulmonary nodule. In one patient (above), the nodule showed no tracer uptake, and a biopsy was avoided. In the other patient (below), the nodule showed avid tracer uptake. Moreover, whole-body PET/CT allowed immediate staging of the patient with the very suspicious lung lesion. Metabolic characterisation of pulmonary nodules is also very important when these are located in areas which are impossible to reach by a biopsy needle (Images courtesy of HSR, Milan)

