The New 24/4 Oral Contraceptive Pill
by Yves Muscat Baron MD PhD FRCOG FRCPI
Consultant Obstetrician and Gynaecologist
Mater Dei Hospital
“It is our experience, as it was our aim, that as a result of child-spacing, and adequate care of mothers, death rates would be reduced. It is now a fact that as a result of birth control, the survival rate among mothers and children is higher. There is less suffering for all groups”
Margaret Sanger, 1947
Historical Background
The above quotation came from Margaret Sanger, then President of the American Society of Family Planning, who had the vision that family planning would holistically be beneficial to society. In fact in 1951, Margaret Sanger through her ardent supporter, Katherine Dexter Mc Cormick, collected a total of $150,000 (a substantial amount for the time) so as to fund research on oral contraception. With this funding Dr Gregory Pincus commenced preclinical trials suppressing ovulation in rabbits. Within a decade Dr Carl Djerassi was able to produce the first oral contraceptive pill which was initially tested in Puerto Rico and Haiti.
Since the 1950’s, gynaecological endocrine research in the form of the combination of a progestogen and an oestrogen, given either for postmenopausal hormone replacement or for contraception, remains the most scientifically studied medical treatment. Moreover these gynaecological endocrine developments have probably been the most socially beneficial medical therapy of all time. Despite over half a century of scientific development, improvements in contraceptive regimens are still being developed.
Introduction
The most recent development is the use of a new progestogen drospirenone, whose antimineralocorticoid and antiandrogenic properties lead to improvements in both physical and psychological well-being. Better progestogens with a longer half-life allow a reduction or even an obliteration of the pill free interval and hence a lessening of menstrual-related side effects.
Progesterone has a high affinity for the mineralocorticoid receptor and is an antagonist of aldosterone. Almost all synthetic progestogens are devoid of an antimineralocorticoid effect, and are unable to antagonize the salt-retaining effect of oestrogens. The results of clinical studies with drospirenone, demonstrate significant antialdosterone activity.
Another new development is the 24/4 extended pill formulation. This entails the reduction in the pill free period from seven to four days. The pill free period was suggested in the early pill regimens in an effort to mirror the body’s physiological hormonal patterns. It may also have been introduced to assuage the prescriber’s ‘clinical conscience’ and to allay his patient’s fears that the body would be given a ‘rest’ from the unnatural hormonal ingestion.
Extended pill formulations have several advantages. It is postulated that in practice an extended pill regimen should compensate for inadvertent pill omission reducing the risk of failed contraception. Premenstrual somatic and psychological symptoms are at their highest during the pill free period. Shortening this pill free period together with a long half-life progestin should be able to reduce these undesirable effects.
Scientific Evidence on the New 24/4 OCP
One of the main causes for failure of oral contraception is lack of compliance. Over fifty percent of women taking the oral contraception discontinue or inadvertently omit its ingestion within the first year of its prescription. Recent studies indicate high user satisfaction with drospirenone 3mg and with both ethinyloestradiol formulations of 30µg and 20µg. In the pivotal trial of Bachmann et al, 86% of women were ‘satisfied’ or ‘very satisfied’ with this treatment. Significant proportions, 37.9% and 29.3% of women, reported that physical and emotional well-being, respectively, improved during treatment versus baseline. Moreover, 72.7% of women would have continued with the study medication if given the choice.
Epidemiological reviews regarding menstruation have shown that the majority of women prefer monthly withdrawal bleeds. Again in the pivotal efficacy study, 24/4 drospirenone 3mg and ethinyloestradiol 20µg formulation, had a positive effect on menstruation in the great majority of women. At baseline, 89.6% had satisfactory withdrawal bleeds and this increased to 94.4% through to cycle 13. The severity of withdrawal bleeding was described as ‘spotting’ to ‘normal’ in ≥88% of women. Bleeding patterns as assessed in a 7-cycle open-label randomized study, in the 24/4 drospirenone 3mg and ethinyloestradiol 20µg regimen [n=229] were lighter than the desogestrel 150µg and ethinyloestradiol 20µg regimen [n=220]. The majority of women (>80% during cycles 2 to 13) experienced no intermenstrual bleeding.1 This impact on menstrual flow may be ideal for women suffering from menorrhagia.
| Symptoms |
Hormone treatment (21 days) % |
Hormone-Free Interval (7 days) % |
P-value |
| Pelvic pain | 21 | 70 | <0.001 |
| Headaches | 53 | 70 | <0.001 |
| Breast tenderness | 19 | 58 | <0.001 |
| Bloating/swelling | 16 | 38 | <0.001 |
| Use of pain medications | 43 | 69 | <0.001 |
Table 1: Menstruation-related symptoms and 21/7 oral contraception
The above table regarding oral 21/7 contraception and menstrual symptoms, clearly shows the significant increase in somatic symptoms during the 7 day hormone free period. It was postulated that reducing the duration of the hormone-free period from 7 to 4 days may influence the above symptoms which are frequently a cause for pill discontinuation.
The physiological rationale for shortening the hormone-free interval resulted in avoiding hormonal fluctuations. It is critical that follicular development is efficiently suppressed so as to avoid endogenous oestradiol production. FSH increases during the pill-free interval leading to follicular growth and oestradiol production. Oestradiol fluctuations have been correlated with oral contraception related side-effects. Moreover many of these symptoms are related to increased water retention.
The 24/4 drospirenone 3 mg and ethinyloestradiol 20µg formulation has been shown to maintain the same concentrations in total body water and extracellular water as observed in the follicular phase. This effect is likely due to the antimineralocorticoid activity of drospirenone, which counteracts the water retention elicited by oestrogen.2 The 30 hour half-life of drospirenone extends its antimineralocorticoid into the shortened ‘hormone free period’ reducing the incidence of menstrual related somatic side-effects. In fact, the 24/4 drospirenone 3mg and ethinyloestradiol 20µg formulation led to significant reductions in breast tenderness, bloating, weight gain and headache by p < 0.0033 and p < 0.0001 in the cross-over study and parallel studies respectively.
The effect on lipid and carbohydrate metabolism and haemostatic factors over 7 cycles of 24/4 3mg drospirenone and 20µg ethinyloestradiol formulation were also assessed. These effects were compared to desogestrel 150µg / ethinyloestradiol 20µg indicating that both treatments increased the beneficial HDL-cholesterol and decreased the deleterious LDL-cholesterol. Haemostatic factors and blood glucose levels measured through glucose tolerance tests remained within normal limits.
Oral contraceptives have long been a useful adjunct in the armamentarium against acne. Two placebo-controlled studies utilizing the 24/4 drospirenone 3 mg and 20µg ethinyloestradiol formulation were conducted in a total of 1072 women complaining of moderate acne over 6 cycles. Both studies resulted in significantly (p<0.0001) greater reductions in mean change from baseline in inflammatory, non-inflammatory and total acne lesion counts versus placebo. These results are likely to have been mediated through the anti-androgenic effects of drospirenone.
Oral contraceptive-induced hypertension occurs in approximately 5% of OCP users. This a well-recognized phenomenon characterized by an increase in blood pressure levels after beginning oestrogen/progestin therapy and a reduction to normal levels within 1 year after suspension of the therapy. The pathophysiology of this condition is still not understood although several lines of evidence indicate a role for a genetic susceptibility to the development of this condition. In postmenopausal women receiving hormone replacement in the form of drospirenone /17-beta oestradiol, blood pressure was reduced by -8.6/-5.8 mm Hg versus -3.7/-2.9 mm Hg in those receiving placebo (P < .01 for both SBP and DBP). In the pivotal efficacy study, blood pressure levels in women taking oral contraception utilizing 24/4 drospirenone 3 mg and 20µg ethinyloestradiol formulation remained within normal limits. The drospirenone component may be protective against oral contraceptive-induced hypertension.
As the efforts of Pincus and Djerassi testified more than 50 years ago, the raison d’etre of the oral contraceptive pill is ovulation suppression with resultant contraception. Out of a total of 2004 woman years of exposure to 24/4 drospirenone / ethinyloestradiol regimen across 4 phase III efficacy and cycle control studies a total of 16 pregnancies were observed. This resulted in a Pearl Index of 0.8 for typical use and calculated to be 0.41 for perfect use. This corresponds to more than 99% contraceptive protection.
The first pill in any oral contraceptive pack is the most important pill in preventing pregnancy. Omission of the first pill increases the likelihood of escape ovulations. Suppression of follicular development as determined by the Hoogland scores (transvaginal ultrasound scoring for follicular assessment ) was more pronounced with 24/4 compared to 21/7, drospirenone /ethinyloestradiol formulations (table 2). In the case of three missed pills, a lower incidence of escape ovulations occurred with the 24/4 regimen compared to the 21/7 regimen (2% versus 8%, respectively). The 24/4 regimen consistently suppressed endogenous estrodiol and consequent FSH and LH levels more than the 21/7 regimen.
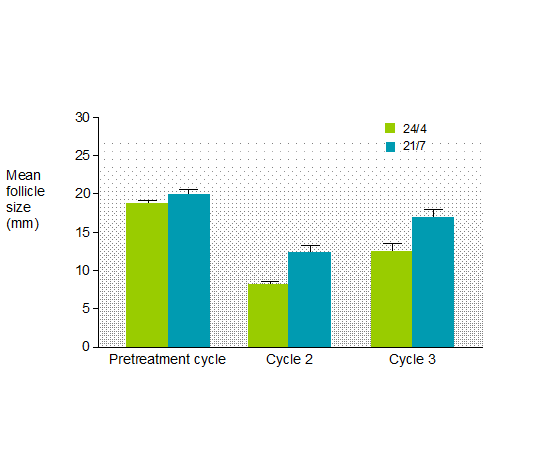
Table 2: Ovulation inhibition study
Conclusion
What would the world be without hormonal contraception? Undoubtedly millions of unplanned pregnancies would have occurred and as a consequence the spectre of abortion, already rife especially in Eastern Europe and the former Soviet Union, would reach catastrophic proportions globally. Noncontraceptive advantages as avidly demonstrated by the 24/4 drospirenone / ethinyloestradiol formulation certainly alleviate the burden on the health services to treat menorrhagia, irregular menstrual bleeding, acne, and the premenstrual syndrome. Epidemiological studies ascertain that without the pill there would be many more deaths from ovarian and endometrial cancer, and from complications following hysterectomy or anaemia. Margaret Sanger’s vision that family planning would impact our social welfare was indeed very close to the truth – “the survival rate among mothers and children is higher. There is less suffering for all groups”.
References
1. Bachmann G, Sulak PJ , Sampson-Landers C et al. Efficacy and safety of a low-dose 24-day combined oral contraceptive containing 20μg ethinylestradiol and 3mg drospirenone. Contraception 2004; 70(3):191–8.
2. Fruzzetti F, Lello S, Lazzarini V et al. The oral contraceptive containing 30μg of ethinylestradiol plus 3mg of drospirenone is able to antagonize the increase of extracellular water occurring in healthy young women during the luteal phase of the menstrual cycle: an observational study. Contraception 2007, 75(3):199–203.
Bibliography
Blode H, Wuttke W, Lock W, Roll G, Heithecker R. A 1-year pharmacokinetic investigation of a novel oral contraceptive containing drospirenone in healthy female volunteers. Eur J Contracept Reprod Health Care 2000; 5(4):256-64.
Fenton C, Wellington K, Moen M, Robinson D. Drospirenone/ethinylestradiol 3mg/20µg 24/4 day regimen: a review of its use in contraception, premenstrual dysphoric syndrome and moderate acne vulgaris. Drugs 2007; 67:1749-65.
Lopez L, Kaptein A, Helmerhorst F. Oral contraceptives containing drospirenone for premenstrual syndrome. Cochrane Database Syst Rev. 2008; 1: CD 6586.
Pearlstein T, Bachmann G, Zacur H, Yonkers K. Treatment of premenstrual dysphoric disorder with a new drospirenone – containing oral contraceptive formulation. Contraception 2005; 72(6):414-21.

