The Mammographically Dense Breast: Strategies in Breast Cancer Imaging
Pierre Vassallo
We have known for some time that a dense breast on mammography presents diagnostic problems in the detection of breast cancer. It has also been suggested that women with dense breasts are at a higher risk for developing breast cancer.
The aim of this article is to update the reader on current thoughts and data on the above issues and discusses the use of supplemental imaging in women with dense breasts, including digital breast tomosynthesis (DBT), whole-breast ultrasonography (WBUS), automated whole-breast US (ABUS), and gadolinium-enhanced breast magnetic resonance (MR) imaging.
Breast density refers to the visual characteristics of the breast on mammograms. Breasts containing a high proportion of parenchymal elements and ducts compared to background fatty tissue are considered dense, while those containing a higher proportion fatty tissue are considered non-dense or fatty in composition. In layman’s terms, a dense breast has more “white” elements that may be patchy or confluent (Fig 1a), while a fatty or non-dense breast contains more “dark” elements (Fig 1b) on a mammogram.
Breast density does not correlate with firmness noted on clinical palpation. A firm breast may have a predominantly fatty composition, while a soft breast may have a dense composition on mammography.
Breast density has been classified into four groups: fatty breasts, scattered fibro-glandular elements, heterogeneously dense breasts and very (or homogeneously) dense breasts (Fig 2). There is significant variation in classification between different observers and even between repeat reviews by the same observer, particularly towards the middle of the scale, due to its non-quantitative nature. There have been attempts to quantify breast density on digital mammograms through software analysis with very limited success.
Data from the Breast Cancer Surveillance Consortium (BCSC) collected from registries around the United States show that on 934,098 normal screening mammograms obtained from 1994 to 2008, the distribution according to tissue density was as follows: 9.0% of breasts were fatty breasts, 44.1% were breasts with scattered areas of fibro glandular densities 38.3% were heterogeneously dense breasts, and 8.6% were extremely (or “homogeneously”) dense breasts. In clinical practice, fatty breasts and those with scattered fibro glandular elements are referred to as non-dense breasts, while those that are heterogeneously or extremely dense are referred to as dense breasts.
Importantly, breast density diminishes with increasing patient age and body mass index. A decrease in blood oestrogen and progesterone levels as seen most notably at menopause, will result in a decrease in glandular and ductal components and a relative increase in fatty components in the breast. Weight-loss or gain will influence the fat content in the breast and will alter breast density (Fig 3).
Higher breast density may obscure a breast cancer during mammographic evaluation and result in later diagnosis when the cancer is more advanced. This observation has led to the introduction of legislative changes in several states in the USA that obliges the radiologist to notify the patient regarding her breast density and the possible need for supplemental imaging.
There is considerable evidence that breast cancers can be obscured by dense breast tissue. Past studies have used the concept of interval cancers to distinguish those cancers that appeared on mammograms obtained within 1 year of the prior mammograms. Non-interval cancers were those detected after a period greater than 12 months from the previous mammograms. It was found that in women with dense breasts, the proportion of interval cancers was significantly higher than in those with non-dense breasts. The incidence of non-interval cancers was higher in women with non-dense breast that in those with dense breasts. This lead to the conclusion that very early signs of breast cancer may have been obscured by abundant parenchymal elements in dense breasts on the initial mammograms.
Other supporting evidence for the obscuring effect caused by dense parenchymal elements has been obtained through the use of supplemental investigations such as breast ultrasound (US), digital breast tomosynthesis (DBT) and breast MRI, which help detect cancers that are not visible in dense breasts. Numerous studies have evaluated the use of supplemental imaging to improve breast cancer detection, however significant differences in study design and patient selection criteria between these studies make it difficult to reach concrete conclusions.
A most significant improvement in the ability to evaluate the dense breast was seen with the introduction of Digital Mammography. The image quality provided by digital mammograms significantly improved visualisation of the dense breast compared to earlier film/screen mammograms. There are two types of digital mammograms: Computed Radiography (CR) Mammograms and Full Field Digital Mammograms (also known as DR Mammograms). DR Mammograms have a much better image quality than CR Mammograms (Fig 4). FFDM is the gold standard for breast cancer screening today.
A standard strategy used by most radiologists when faced with a mammogram showing dense breasts is to proceed to a breast ultrasound (or WBUS) to obtain a second view of those difficult-to-analyse areas on the mammogram. Breast ultrasound does not rely on tissue density to create an image, it relies on tissue and cell interfaces that act as acoustic reflectors. What is seen a dense on mammography is usually well penetrated by the ultrasound beam. Mammographically difficult areas in the breast are usually cleared with a breast ultrasound often performed in the same sitting (Fig 5) providing an efficient and effective mode of patient management.
Breast ultrasound is widely available and inexpensive, but it is strongly both operator and technology dependent. The abundance of older ultrasound machines in many hospitals and radiology practices and the lack of trained expertise would significantly reduce its value for breast cancer detection. The approval of ABUS but the FDA (Food and Drug Administration, USA) in 2011, may take a step towards improving the situation. ABUS takes a mechanically-controlled scan through the whole breast and records a 3D image for review by the radiologist. The inherent advantage of free-hand breast ultrasound is that the examiner is able to rotate the probe to select the best plane of lesion visibility. This is not possible with ABUS. ABUS has not been approved for breast cancer screening.
DBT is a new technology based on DR Mammography, which obtains a series of images at different levels through the breast similar to the consecutive slices obtained with Computed Tomography (CT). Sometimes referred to as 3D mammography, it is not a 3D imaging method as the thickness of the slices obtained is too large to allow 3D reconstructions. DBT appears to be of value in the dense breast as it reduces lesion superimposition by dense parenchymal structure (Fig 6). However, DBT is used as an adjunct to standard DR mammograms with the consequence that there is a doubling of radiation exposure to the patient if DBT is performed in just one plane. DBT is therefore not recommended in this format as a screening method for breast cancer. It can be used to evaluate patients with abnormal mammograms or those with significant clinical suspicion. The possibility to create synthesised DR mammograms from DBT data would reduce the radiation exposure by avoiding initial DR Mammography and may in future lead DBT becoming a primary screening tool.
MR Imaging of the breast is the most sensitive test available for detecting breast cancer. It will depict more cancers that DR Mammography or Breast Ultrasound, but it also has a higher false positive rate, which would result in unnecessary breast biopsies. There are clear guidelines that recommend the use of breast MR imaging for screening women with a high risk for breast cancer. Whether it should also be used for intermediate or low risk women is as yet undecided. Breast MR imaging depends on morphological features and contrast enhancement characteristics to detect cancerous lesions in the breast (Fig 7). Long examination times, limited scanner availability, high cost and the need for intravenous injection of contrast material limit the use of MR imaging for breast cancer screening.
A further question relating to the dense breast is whether breast density in itself makes the patient more prone to breast cancer. This is a subject under constant debate. The higher volume of glandular and ductal elements in dense breasts, which are the tissues that give rise to breast cancer, may raise the patient’s risk. There is some statistical evidence that breast density may influence a patient’s risk to develop breast cancer, however other risk factors such as positive breast cancer gene testing, family and personal history and obesity and smoking play a greater role.
The dense breast poses a very frequent problem encountered in the breast cancer screening with mammography. The use of supplemental imaging, mainly breast ultrasound and MR imaging, significantly improves the accuracy of diagnostic assessment in the mammographically dense breast. Finally, correlation of clinical with imaging data is of great importance as clinical data may provide helpful guidance in those difficult areas in the dense breast.
Figure Legends
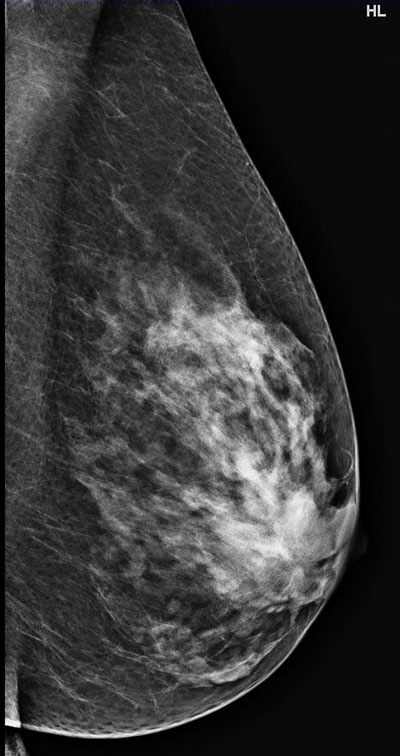
Figure 1. Mammograms showing a heterogeneously dense breast (a)
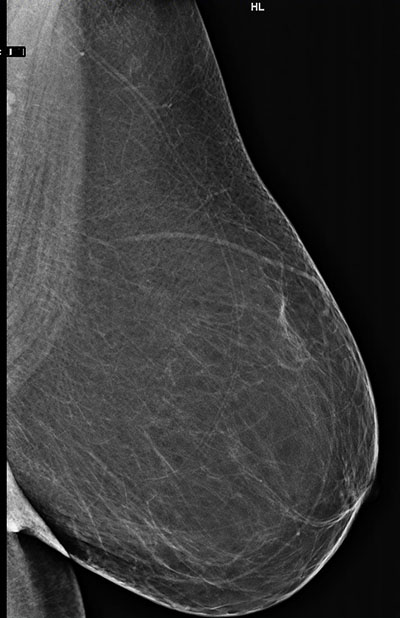
compared to a fatty breast (b).
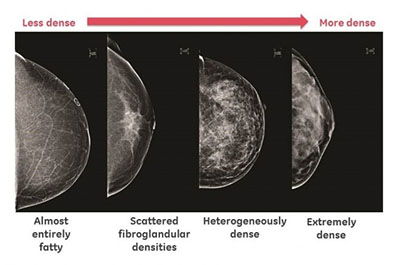
Figure 2. Grading of breast density on a mammogram.
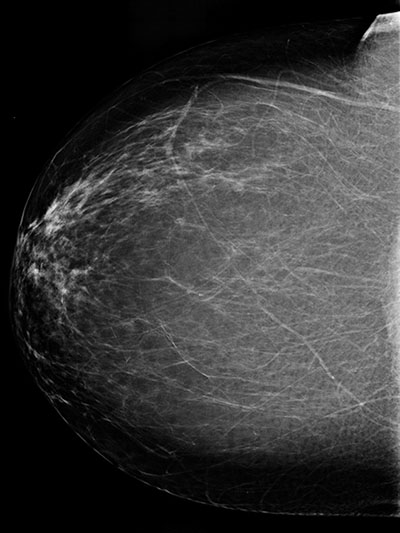
Figure 3. Mammograms of the same patient taken 2 years apart, (a) before and
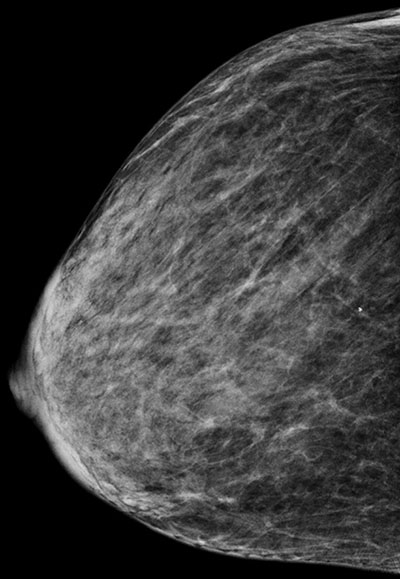
(b) after considerable weight loss.
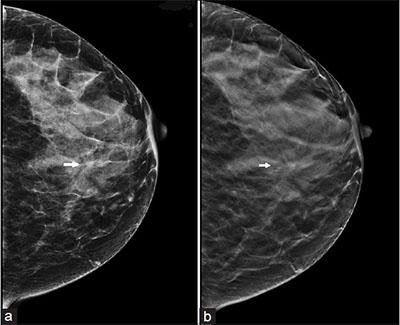
Figure 4. DR (a) vs CR (b) Mammograms: The same lesion (arrow) is more clearly depicted on the DR mammogram than on the CR mammogram.
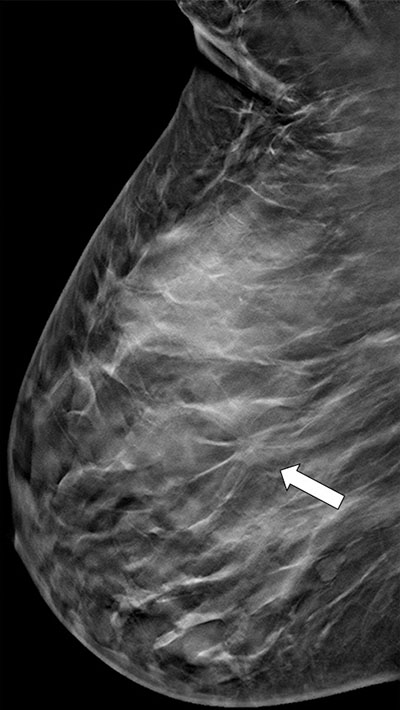
Figure 5. Suspected lesion (arrow) on mammogram (a)
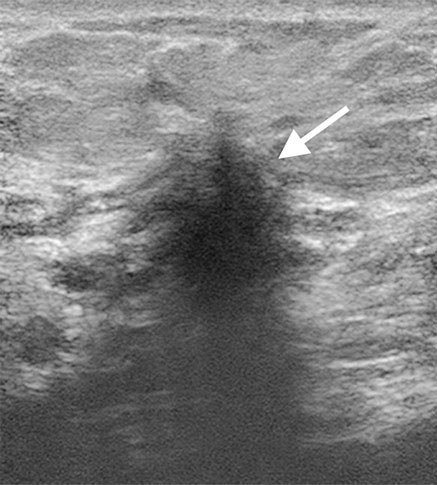
was clearly confirmed on breast ultrasound (b).
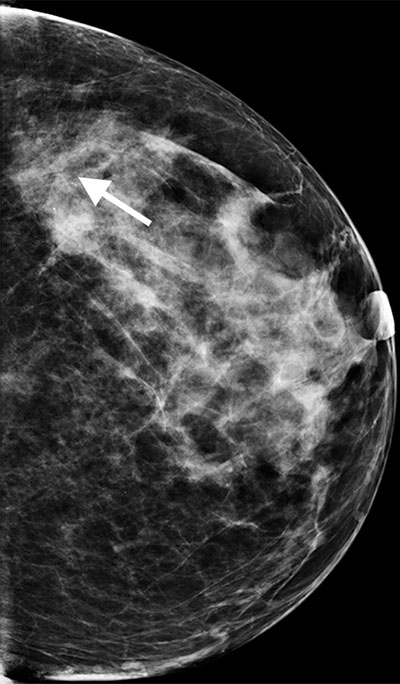
Figure 6. Subtle lesion (arrow) seen on DR mammogram (a)
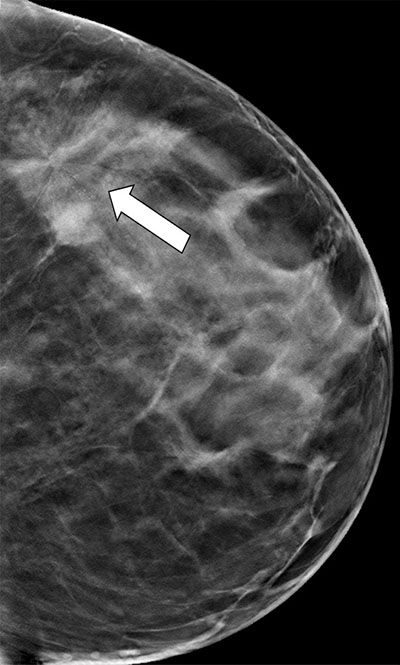
better seen on supplemental DBT (b).
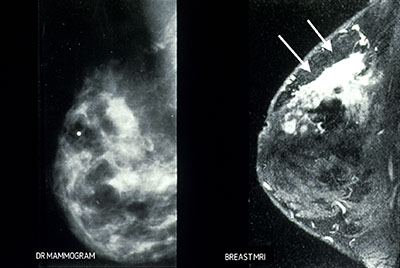
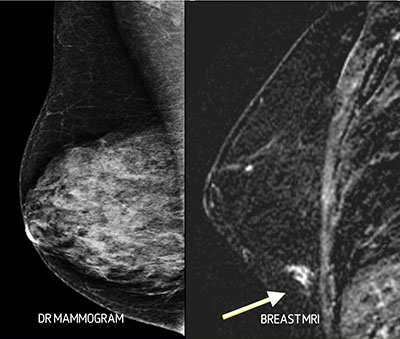
Figure 7. Two cases of cancer (a and b) obscured in dense breasts that were visible (arrows) on MR imaging.

