Targeting chemokines: new drugs for old diseases
by Anthony G Fenech* B.Pharm.(Hons) MPhil PhD (Nott)
Department of Clinical Pharmacology and Therapeutics
Faculty of Medicine and Surgery
University of Malta
Email: anthony.fenech@um.edu.mt
Michael A Portelli B.Pharm.(Hons)
* corresponding author
Chemokine receptor antagonists are set to become novel important pharmacological tools within the current therapeutic repertoire available for the management of various inflammatory conditions. Currently in various phases of drug development, these molecules act by inhibiting specific inflammatory pathways, and current research suggests a safer adverse effect profile for these drugs than today’s therapy.1
Introduction
Chemokines represent a large family of proteins that are produced by a variety of cells, including stromal cells, epithelial cells and immune cells. They are cell activators and chemoattractants, regulating the traffic of cells through a chemotactic gradient. They have a pivotal role in the trafficking of a variety of leukocytes such as T-lymphocytes and neutrophils and endothelial cells: (a) within primary lymphoid organs, (b) from primary to secondary lymphoid organs, (c) between secondary lymphoid organs, and (d) during a humoral immune response.2 They therefore posses an essential role in both the inflammatory and immunological processes.
Classification
Over forty human chemokines have been currently identified (Table 1), and these have been classified by structure into four sub-classes, according to the number and spacing of conserved cysteine residues in the chemotactic domain. These four sub-classes are given the preferred names C, CC, CXC and CXXXC, with CC possessing two cysteine residues adjacent to each other, CXC and CXXXC possessing one and three amino acids respectively between the cysteine residues and the C having one of the two cysteine residues missing (Figure 1).
| Chemokine | Former designation | Receptor(s) |
| CCL3 | MIP-1α | CCR1, CCR5 |
| CCL5 | RANTES | CCR1, CCR3, CCR5 |
| CCL8 | MCP-2 | CCR1, CCR2, CCR3 |
| CCL17 | TARC | CCR4 |
| CXCL12 | SDF-1, PBSF | CXCR4 |
Table 1. Some chemokine ligands and their specific receptors
MIP-1α : Macrophage inflammatory protein 1 alpha, RANTES: Regulated upon Activation, Normal T-cell Expressed, and Secreted, MCP-2: Macrophage chemoattractant protein 2, TARC: Thymus and activation-regulated chemokine, SDF-1: stromal cell-derived factor-1, PBSF: pre-B cell growth-stimulating factor.
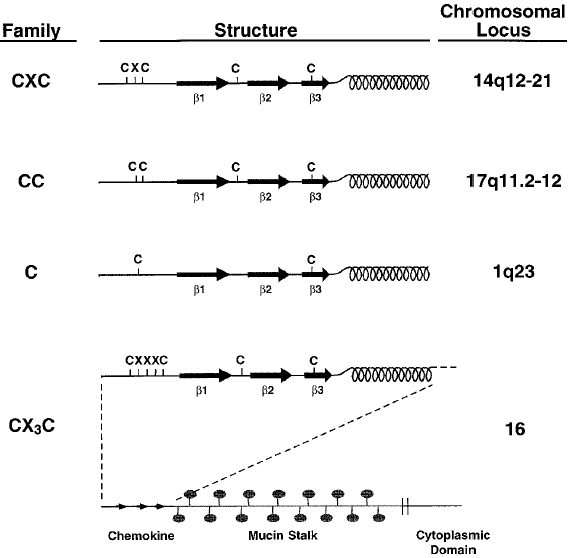
Fig 1 Chemokine families.
The common structure and chromosomal location of the four chemokine sub-classes, showing the location of cysteine residues, after which they are named. All chemokines have at least three β-pleated sheets (indicated as β1-3) and a C-terminal α-helix. The CX3C chemokine domain occurs at the end of a long stalk which is heavily substituted with mucin-like carbohydrates. The protein is embedded in the membrane and has a short cytoplasmic domain.
(Reproduced from Rollins, 1997.22)
Chemokine receptors
Chemokine receptors are structurally and functionally related proteins, that are members of the class A G-protein coupled receptors (GPCRs); they are all coupled to Ginhibitory proteins.3
Currently 18 proteins that meet the definition for a chemokine receptor exist, with each sub-family of the chemokine class of inflammatory mediators having its own sub-class of receptors. Of these 18 known proteins, 10 are known to be CCRs, 6 are CXCRs, and only one receptor is known for each of the CXXXC and C sub-classes (CXXXCR1 and XCR1 respectively). Of all of these receptors, it is of interest to note that only the CXCR4 has been found to be essential for life.4
Chemokine receptors are commonly known to exhibit promiscuity,5 that is, while certain chemokines show a preference for their own specific receptor; they may also interact with other receptors, causing activation. With the same idea, receptors themselves may also be activated by other, non-specific chemokines. This is known as the shared receptor concept, which occurs within, but not between the CC or CXC branches, except for the Duffy antigen/receptor for chemokine (DARC) which binds to the CC and CXC classes with equal affinity.6 An example of this concept is the chemokine ligand MIP-1α which binds with high affinity to CCR1, CCR3 and CCR5.
Mode of action
Chemokines act by regulating leukocyte recruitment; causing cellular activation and inflammatory mediator release by promoting the Th2 inflammatory response and by regulating the release of IgE. Their immunomodulatory and chemoattractant action occurs via the infiltration of inflammatory cells and by the inflammatory burst caused from eosinophils and basophils. This causes the migration of leukocytes, a critical component of systemic immunity, from the vascular compartment into the tissues through a process of chemotaxis. This highlights the importance of chemokines as inflammatory mediators as well as the significance of their receptors, the regulation of expression of which is recognised as one of the key events in leukocyte localisation and trafficking during inflammation. Indeed, chemokine expression has been reported to play a major role in (a) glomerulonephritis, (b) inflammatory bowel disease, (c) allergenic transplant rejection, (d) atopic dermatitis, (e) allergic rhinitis, (f) asthma, and (g) chronic obstructive pulmonary disease.7 This list further includes auto-immune disease such as diabetes and multiple sclerosis, stemming from the fact that chemokines are also able to direct immune cells to target and destroy perfectly healthy cells.
Therefore antagonism of various chemokine receptors has been proposed as a useful therapeutic approach in the management of inflammatory conditions, and indeed several such molecules are currently under development as novel therapeutic agents.8
Chemokine receptor antagonists
The shared receptor concept may make it more beneficial for the pharmaceutical industry to design and produce receptor antagonists, rather than drugs that antagonise the chemokine ligands directly. Indeed various chemokine receptor antagonists are currently being developed as novel therapeutic tools for a variety of diseases. For example, CCR4 antagonists are currently under development for asthma therapeutics. High levels of the CCR4 ligands RANTES, MCP-1, TARC and MDC have been detected in both the sputum and serum of asthmatics.9 Antagonism of the CCR4, a receptor that has been determined to be a selective marker for Th2 type lymphocytes,5,10 has been shown to result in decreased eosinophilia and diminished airway hyperresponsivness.11,12
It has been recently demonstrated that CCR4 antagonism may be used as a mechanism to inhibit the recruitment of activated leukocytes.13 These CCR4 antagonists include the neutralising antibody 1G1, which has been shown to potentially block TARC binding to CCR4 transfectants and to prevent chemotaxis of these cells in response to MDC and TARC.7 Also under development are spironolactams and spiroperidines,3 a series of 2-aminothiazole derivatives aimed at inhibiting TARC and MDC binding to the CCR4,14 as well as 2,4-diaminoquinazolines, whose administration in a murine model of acute dermatitis showed significant anti-inflammatory activity.15
Other antagonists include the pyrazolone methylamino piperidine derivatives and the patented product GW-766994, both CCR3 antagonists, the latter of which is currently undergoing Phase II clinical trials for the treatment of asthma and allergic rhinitis. Besides having CCR4 antagonistic activity, the spiroperidines also exert a very potent CCR2 blocking action,16 while the CCR1 antagonist BX471 has been reported to be efficient in a rat heterotrophic heart transplant rejection model.17
Conclusion
Chemokine receptor antagonists therefore have a future in the treatment of inflammatory disease, not only since they seem to modify disease with minimal impact on normal host defence, but also since their action seems to be highly specific with few adverse effects.18
The shared receptor concept approach requires the development of antagonists that target a wide variety of receptors at the same time, whose potency would be dependent on the affinity to the receptor and its degree of antagonistic effect. This would avoid the use of complex antagonist mixtures.
Besides the future of chemokine receptor antagonists as anti-inflammatory agents and immunosupressants, one must not fail to mention the advances other types of chemokine antagonists have had in other therapeutic fields. Chemokines have been found to be used by various intracellular pathogens, most notably HIV and Plasmodium vivax to facilitate their entry and transmission, and therefore they may represent novel antiviral and antiparasitic treatment. Indeed, TAK-779, the first reported non-peptide antagonist19 is a CCR5 antagonist that has recently been in Phase I clinical trials as an anti-HIV treatment.20 More recently reported CCR5 antagonists include 873140 (aplavira) and UK 427857 (maraviroc),21 approved by the European Medicines Agency in September 2007, and SCH 351125 (vicriviroc), currently undergoing Phase II clinical trials,22 all of which contribute to the antiviral therapeutic repertoire.
References
1. Chemokine Therapeutics Fact Sheet. Available at http://www.chemokine.net/fact_sheet.htm.
2. Melchers F, Rolink AG, Schaniel C. The Role of Chemokines in Regulating Cell Migration during Humoral Immune Responses. Cell 1999; 99:351-4.
3. Pease JE, Williams TJ. Chemokines and their receptors in allergic disease. J Allergy Clin Immunol 2006; 118:305-18.
4. Murphy PM, Baggiolini M, Charo IF et al. International Union of Pharmacology. XXII. Nomenclature for Chemokine Receptors. Pharmacol Rev 2000; 52:145-76.
5. Smit JJ, Lukacs NW. A closer look at chemokines and their role in asthmatic responses. Eur J Pharmacol 2006; 533:277-88.
6. Horuk R. Chemokine receptors. Cytokine Growth Factor Rev 2001; 12:313-35.
7. Chantry D, Burgess LE. Chemokines in Allergy. Curr Drug Targets Inflamm Allergy 2002; 1:109-16.
8. United States Trademark and Patent Office. Available at: http://www.uspto.gov/
9. Sekiya T, Yamada H, Yamaguchi M et al. Increased Levels of a TH2-type CC Chemokine Thymus and activation regulated chemokine (TARC) in serum and induced sputum of asthmatics. Allergy 2002; 57:173-7.
10. Peroni G.D, Bordignon PP, Piacenti L.G et al. CC chemokine receptor expression in childhood asthma is influenced by natural allergen exposure. Pediatr Allergy Immunol 1996; 17:495-500.
11. Gonzalo JA, Pan y, Lloyd CM et al. Mouse monocyte-derived chemokine is involved in airway hyperreactivity and lung inflammation. J Immunol 1999; 163:403-11.
12. Kawasasaki S, Takizawa H, Yoneyama H et al. Intervention of thymus and activation regulated chemokine attenuates the development of allergic airway inflammation and hyperresponsivness in mice. J Immunol 2001; 166:2055-62.
13. Purandare AV, Wan H, Gao A et al. Optimization of CCR4 antagonists: Side-chain exploration. Bioorg Med Chem Lett 2006; 16:204-7.
14. Wang X, Xu F, Xu Q et al. Optimization of 2-aminthiazole derivatives as CCR4 antagonists. Bioorg Med Chem Lett 2006; 16:2800-3.
15. Yokoyama K, Ishikawa N, Igarashi S et al. Potent CCR4 antagonists: Synthesis, evaluation, and docking study of 2,4-diaminoquinazolines. Bioorg Med Chem Lett 2008; 16(17):7968-74.
16. Cascieri MA, Springer MS. The chemokine/chemokine-receptor family: potential and progress for therapeutic interventions. Curr Opin Chem Biol 2000; 4:420-7.
17. Horuk R, Clayberger C, Krensky AM et al. A non-peptide functional antagonist of the CCR1 chemokine receptor is effective in rat heart transplant rejection. J Biol Chem 2000; 275:25562-71.
18. Power CA, Proudfoot AE. The chemokine system: novel broad-spectrum therapeutic targets. Curr Opin Pharmacol 2001; 1:417-24.
19. Lagu B, Gerchak C, Pan M et al. Potent and selective CC-chemokine receptor-2 (CCR2) antagonists as a potential treatment for asthma. Bioorg Med Chem Lett 2007; 17:4382-6.
20. Hayworth B, Lin H, Fidock M, Dorr P, Strange PG. Allosteric effects of antagonists on signaling by the chemokine receptor CCR5. Bio Pha 2007; 74:891-7.
21. MacArthur RD, Novack RM. Reviews of anti-infective agents: maraviroc: the first of a new class of antiretroviral agent. Clin Infect Dis 2008; 47:236-41.
22. Rollins BJ. Chemokines. Blood 1997; 3:909-28.

