Imaging Prostate Cancer
Dr Pierre Vassallo
Prostate cancer is the most common cancer in men.1 It normally presents a long slow course with few or no symptoms. Consequently, in 17% of cases, metastatic disease is already present at the time of initial diagnosis. It shows great variability in its biological and clinical behaviour ranging from indolent localised disease to highly aggressive disease with metastases.
The role of Prostate Specific Antigen (PSA) testing
The screening test used for detecting prostate cancer is total serum prostate-specific antigen (PSA) measurement. Normal serum PSA levels increase with age. The standard normal total serum PSA level is 0.0-4.0ng/mL. However, the age specific ranges are as follows: 0-2.5ng/mL for age 40-49 years, 0-3.5ng/mL for age 50-59 years, 0-4.5ng/mL for age 60-69 years and 0-6.5ng/mL for age 70-79 years.
Individuals with total serum PSA levels of 4-10ng/mL should be further investigated with a serum free PSA to total PSA ratio; free PSA levels >20% are indicative of benign prostatic disease, while levels <10% are more suggestive of prostatic malignancy. A total serum PSA level >10ng/mL is highly suggestive of prostate cancer with higher levels suggesting metastatic spread.
PSA is also monitored after definitive treatment for prostate cancer since a rising PSA level is usually indicative of recurrent prostate cancer. The PSA level should be measured every 6-12 months for 5 years and yearly thereafter.2
Shortly after radical prostatectomy, PSA levels should be undetectable (<0.1ng/mL). Persistently detectible PSA levels usually indicate residual viable tumour. PSA level recurrences occur in 20-35% of cases usually within 2-3 years after surgery.3
After radical prostatectomy, biochemical recurrence is defined as either two subsequent rises in PSA level after an initial undetectable post-prostatectomy PSA level, or two post-operative readings >0.2ng/mL.
After radiation therapy or tumour ablation, it is normal to have some residual PSA level even in cured patients. However, a rise of >= 0.2ng/mL above post-treatment levels is considered indicative of recurrence.
The median time for development of metastatic disease after biochemical recurrence is 8 years. Thus, the decision whether to treat or not and which form of treatment to implement will depend on the individual’s health and life expectancy as well as prostate cancer-specific clinical, pathologic, radiologic, and genomic factors. Treatment will depend on whether disease is local, metastatic or both.
Imaging Prostate Cancer
Imaging of the prostate is generally advocated within the clinical setting of a rising level of serum PSA.
Conventional imaging including computed tomography (CT) (Fig 1) and Technetium 99m (99Tc)-labelled methylene diphosphonate (MDP) bone scanning (Fig 2) are used for initial staging to detect metastatic disease. Metastatic disease occurs mainly to the bones and lymph nodes; metastases may also be found in the liver, lungs, pleura, adrenal glands, ureter, peritoneum, penis, testes, and meninges.
Increased radiotracer uptake on 99Tc-MDP scans reflects increase bone turnover, which may be caused by metastatic prostate cancer. However increased bone turnover may also occur with fractures, degenerative joint disease, and Paget’s disease. In equivocal 99Tc-MDP scans, further evaluation with skeletal radiography, CT, Magnetic Resonance Imaging (MRI) and biopsy may be required to confirm or exclude metastatic prostate cancer.
CT scanning is useful for monitoring enlarged lymph nodes, sclerotic bone metastases and visceral metastases, while MRI and 99Tc-MDP scans are superior for monitoring bone metastases.4 However, metastatic prostate cancer is often present in normal sized lymph nodes, which are missed on CT scans.5
CT and bone scanning are not useful for assessing the extent of local disease within the prostate and for detecting direct extracapsular extension.
Multiparametric Magnetic Resonance Imaging (MP-MRI) of the prostate is an established imaging method for assessment of loco-regional disease and for detection of recurrence within the prostate or the prostate bed (after prostatectomy).6 MP-MRI is also useful for imaging targeted biopsy of the prostate.
Reporting of MP-MRI findings is done using a structured reporting system called Prostate Imaging – Reporting and Data System (PI-RADS). This system takes all findings seen on different MRI imaging sequences and combines them into a score that is used to guide treatment. PI-RADS scoring is based on findings seen on T2-weighted (T2-w) images, diffusion-weighted images (DWI) and dynamic contrast-enhanced (DCE) T1-weighted images. PI-RADS scores indicate the likelihood of significant cancer and range from PI-RADS 1 (very low) to PI-RADS 5 (very high). Biopsy should be considered with PI-RADS 4 or 5 scores, and observation with PI-RADS 1 or 2 lesions.
On T2-w images, prostate cancer appears as an area of low signal (Fig 3a), while on DWI it shows high signal (Fig 3b). DWI images are used to derive Apparent Diffusion Coefficient (ADC) maps that are used to detect areas of restricted diffusion associated with high cellularity that is seen in malignant lesions; suspicious lesions are seen as dark areas on ADC maps (Fig 3c). DCE imaging is used in combination with T2-w and DWI. If a lesion noted on T2-w or DWI enhances, even if the enhancement is similar to adjacent normal prostatic tissue, this is considered indicative of viable tumour (Fig 3d).
Extracapsular infiltration of prostatic cancer and lymph node metastases are also detected on MP-MRI (Fig 4).
Positron Emission Tomography (PET)/CT and PET/MRI are increasingly used for detection of local and metastatic prostate cancer. Initially radiotracers containing carbon-11 choline and fluorine-18 fluciclovine were used with PET/CT and PET/MRI with some success at detecting foci of prostate cancer. More recently gallium-68 prostate-specific membrane antigen (PMSA) radioactive PET tracers are being implemented to detect local and metastatic prostate cancer (Fig 5). These radiotracers show increased sensitivity and specificity for detecting metastatic disease such as small-volume lymph node metastases.
Conclusion
As is evident above, imaging of prostate cancer is a complex process, where numerous parameters are available that need to be combined to guide effective treatment. However, treatment decisions also depend on other factors including patient age, changing PSA levels, clinical history, local expertise and patient preferences.
Figure Legends
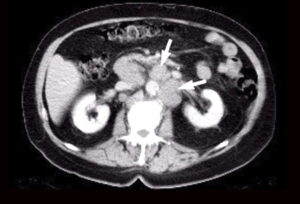
Fig 1. CT scan showing multiple enlarged retroperitoneal lymph nodes (arrows) due to spread of prostate cancer to the lymph nodes.
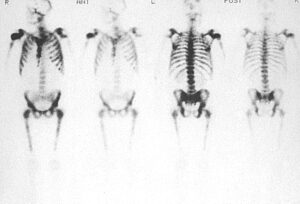
Fig 2. Tc-99m radionuclide bone scan showing marked radiotracer uptake in the spine, ribs, sternum pelvic bones and femora; this is known as a “superscan” appearance. Note that there is no renal excretion or soft tissue uptake of radiotracer due to avid uptake in bone.
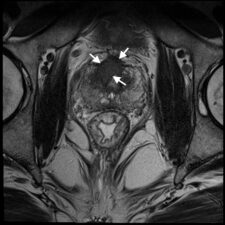
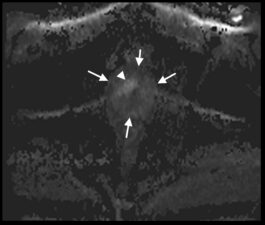
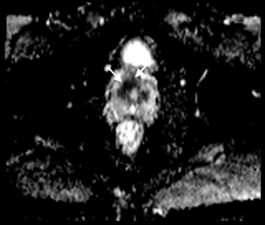
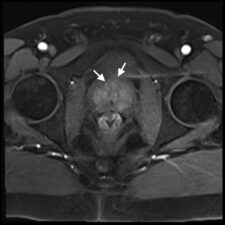
Fig 3. a. T2-w transverse image of the prostate showing an anterior (PI-Rads 5) tumour (arrows) as an area of low signal. b. Transverse DWI image showing the prostate (arrows) and the same tumour as in a that exhibits high signal. c. ADC map showing the same lesion as a dark area (arrows) confirming restricted diffusion. d. Transverse DCE image showing enhancement (arrows) that occurs contemporaneously with surrounding normal prostatic tissue (Case courtesy of Dr Matthew Bastian-Jordan, Radiopaedia.org, rID: 46481).
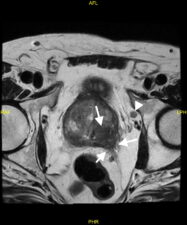
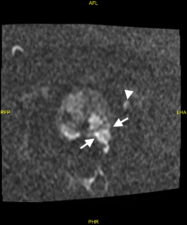
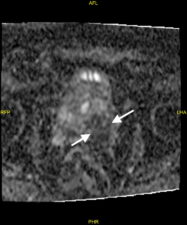
Fig 4. a. T2-w image showing tumour extending through the postero-lateral prostatic capsule (arrows) and an enlarged lymph node (arrowhead). b. The prostatic lesion (arrows) and enlarged lymph node (arrowhead) show high signal in the DWI image. c. The prostatic tumour shows diffusion restriction (low signal – arrows) on the ADC map (Case courtesy of Dr Ian Bickle, Radiopaedia.org, rID: 84984).
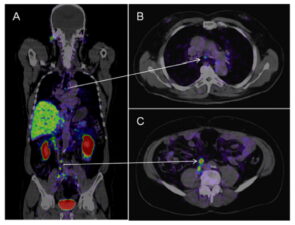
Fig 5. CT/PET Ga68 PMSA scan showing metastatic disease in small lymph nodes in the mediastinum (arrow A and B) and in the retroperitoneum (arrow A and C).
References
- Siegel RL, Miller KD, Jemal A. Cancer statistics, 2020. CA Cancer J Clin 2020;70(1):7-30.
- Mohler JL, Antonarakis ES, Armstrong AJ, et al. Prostate Cancer, Version 2.2019, NCCN Clinical Practice Guidelines in Oncology. Journal of the National Comprehensive Cancer Network : JNCCN 2019;17(5):479-505. ;
- Toussi A, Stewart-Merrill SB, Boorjian SA, et al. Standardizing the Definition of Biochemical Recurrence after Radical Prostatectomy-What Prostate Specific Antigen Cut Point Best Predicts a Durable Increase and Subsequent Systemic Progression? J Urol 2016;195(6):1754-9.
- Hricak H, Schöder H, Pucar D, Lis E, Eberhardt SC, Onyebuchi CN, Scher HI. Advances in imaging in the postoperative patient with a rising prostate-specific antigen level. Semin Oncol 2003;30(5):616-34.
- Fortuin AS, Deserno WM, Meijer HJ, et al. Value of PET/CT and MR lymphography in treatment of prostate cancer patients with lymph node metastases. Int J Radiat Oncol Biol Phys 2012;84(3):712-8.
- Tanaka T, Yang M, Froemming AT, et al. Current Imaging Techniques for and Imaging Spectrum of Prostate Cancer Recurrence and Metastasis: A Pictorial Review. Radiographics 2020;40(3):709-726.

