Imaging Oesophageal Cancer
Pierre Vassallo
The prevalence of oesophageal cancer has increased dramaticallyin the past 30 years, with increases of 350%–800% reportedduring this period. Oesophagealcancer is often detected late with 75% of patients having diseased lymph nodes at initial diagnosis. The 5-year survival rate is only 3% for patients with lymph node involvement,whereas it is 42% for patients who do not have nodal involvement.Approximately 18% of patients will have distant metastases; of these 45% will be to abdominal lymph nodes, 35% to the liver, 20% to the lungs, 18% to supraclavicular nodes, 9% to bone, and 5% to adrenalglands. Consequently, the prognosis is poor, with surgicalcure achieved in less than 10% of patients. Surgeons differin their approach to patients with advanced-stage disease. Palliativetherapies include surgery, laser resection, radiation therapyor chemotherapy, and oesophageal stent placement or dilation.Surgery, whether curative or palliative, carries a significantrisk of mortality that ranges from 5% to 20% depending on thesurgeon’s experience. Therefore, accurate preoperativestaging, particularly with regard to depth of wall invasion,mediastinal invasion, nodal involvement, and distant metastases,is vital in determining the most appropriate therapy and inhelping avoid inappropriate attempts at curative surgery.
Clinical Presentation:
Dysphagia is the most common presenting complaint inpatients with oesophageal cancer, however it is also present in patients with benign oesophageal strictures. The duration of dysphagiais a useful clinical parameter for differentiating benign frommalignant strictures. In general, benign strictures are associatedwith long-standing, intermittent, nonprogressive dysphagia,whereas malignant strictures are associated with recent onsetof rapidly progressive dysphagia and weight loss.
Chronic or severe esophagitis from a variety of causes may leadto scarring and fibrosis with the development of esophagealstrictures. Therefore, the clinical setting is crucial in determiningthe underlying cause of these strictures. In some cases, thecorrect diagnosis may be suggested by a temporal relationshipbetween stricture formation and precipitating factors such asmediastinal irradiation, ingestion of caustic substances, andnasogastric intubation. In other cases, important clinical cluesmay be provided by findings such as high serum gastrin levelsin Zollinger-Ellison syndrome, an allergy history or peripheraleosinophilia in eosinophilic esophagitis, or bullous skin eruptionsin epidermolysis bullosa dystrophica or benign mucous membranepemphigoid. Therefore, all strictures should be evaluated inthe clinical context in which they develop.
Imaging Methods:
Oesophageal strictures are best evaluated with biphasic oesophagography(aka Barium Swallow) that includes both double-contrast and single-contrast spot X-rayimages. The single-contrast phase optimises distension ofthe oesophagus, thereby improving detection of strictures, whereasthe double-contrast phase optimises visualization of the mucosafor nodules, ulcers, or other radiographic findings associatedwith these strictures.
Reflux-induced (gastro-oesophageal reflux disease – GORD) strictures classically appear as smooth,tapered areas of concentric narrowing in the distal oesophagusand range from 1 to 4 cm in length (Fig 1). Other pepticstrictures may be associated with a focal cluster of oesophagealintramural pseudodiverticula (Fig 1).However, asymmetricscarring can lead to asymmetric narrowing and may resemble malignant strictures (Fig 2).
Some patients may have a very short segment of ring-like narrowingat the gastro-oesophageal junction above a hiatal hernia (Fig 3). This may resemble a Schatzki ring (Fig 4), which is a normal oesophago-gastric sphincter. Schatzki rings usually appear as smooth, symmetricring-like constrictions with abrupt borders and a length of only1–3 mm, whereas annular peptic strictureshave more tapered borders and a length of over 4 mm.
Hiatal hernias are seen at barium examination in more than 90%of patients with peptic strictures, so that the possibilityof malignant tumor should be considered when a distal oesophagealstricture is detected in the absence of a hernia. Nevertheless,malignant strictures usually have more irregular and nodularcontours and more abrupt or “shouldered” proximal and distalmargins than do benign peptic strictures (Fig 5).
Barrett esophagus is an acquired condition in which there isprogressive columnar metaplasia of the distal oesophagus as aresult of chronic gastroesophageal reflux and reflux esophagitis. Barrett oesophagus is only detected by oesophagoscopy and requires confirmation by endoscopic biopsy. Because Barrett esophagus is a premalignant conditionassociated with an increased risk of developing oesophageal cancer,many investigators advocate endoscopic surveillance of patientswith known Barrett esophagus to detect dysplastic or early carcinomatouschanges before the development of overt carcinoma.
Spiral Computed Tomography (CT) determines the extent of oesophageal cancer based on morphologic features such as direct infiltration of adjacent structures and on the size of mediastinal and coeliac lymphnodes. Nodes measuring 10mm or larger are considered malignant while smaller ones are deemed reactive, however this threshold is only based on statistical analyses and consequently smaller nodes may contain metastases and larger ones may be reactive. CT can also detect metastases to other structures such as the lungs, liver and bone.
Most malignant tumours metabolise glucose at a much higher ratethan normal tissue; as a result, there is an increased accumulationof the glucose analog 2-[fluorine-18]fluoro-2-deoxy-d-glucose(FDG) in malignant tissue. Positron emission tomography (PET)provides diagnostic information based on this increased FDGuptake and may demonstrate early-stage disease before anystructural abnormality is evident. Studies have shown that 90% of oesophageal cancers demonstrate avid FDG uptake (Fig 7), while normal or inflamed gastric mucosa may demonstratesignificant FDG uptake and may be difficult to differentiatefrom tumors. Therefore PET-FDG is not ideal for the detection of the primary tumour in oesophageal cancer due to lack of specificity and the financial expense of the test. Furthermore, FDG PET isnot helpful in detecting local invasion by the primary tumor or involved local lymph nodes due to its limited intrinsic spatial resolution, which is approximately5 mm (Fig 8).
The major advantage of FDG PET over anatomic imaging modalitiesis its ability to detect distant metastases. The limited spatial resolution of FDG PET is not a problem with metastases that are distant from the primary tumour and from sites of normal increased uptake. Metastasesto the liver, lungs, and skeleton can readily be identifiedat FDG PET (Fig 9). Involvement of the supraclavicular, cervical, and celiac nodes by oesophageal cancer is considered distant metastasis (M1) rather than nodal metastasis(N1) and precludes curative surgery; FDG PET can also detect disease at these sites. Thus an FDGPET covering the whole body (skull base to pelvis) can improve the ability to classify diseaseas either resectable or unresectable based on the presence of distant metastases. CT ormagnetic resonance (MR) imaging may at times be unable to distinguishpostoperative scar from tumor recurrence. FDG PET may be useful in this setting and for monitoring response to radiotherapy or chemotherapy.
In summary, oesophageal cancer is usually detected by oesophagography and oesophagoscopy. Accurate staging of oesophageal cancer is crucial for therapeutic planning and is best done with Spiral CT, which can assess resectibility and can provide a baseline study to assess subsequent response to therapy. FDG PET is useful as an adjunct to Spiral CT for detecting distant metastatic disease and for distinguishing scar from recurrent tumour when CT findings are equivocal.
Figure Legends
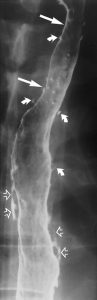
Figure 1. Peptic stricture (large arrow) above a hiatal hernia with oesophageal intramural pseudodiverticulae (small arrows) some forming track like structures (open arrows).
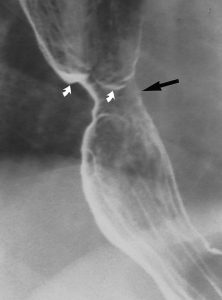
Figure 2. Asymmetric peptic stricture (arrow) due to scarring from reflux esophagitis (white arrows indicate wide pseudodiverticulae).

Figure 3. Ring-like peptic stricture (arrows) above a hiatal hernia, resembling a Schatzki ring, but more asymmetric, having more tapered borders and a greater length than do most Schatzki rings.
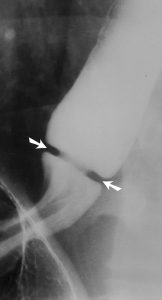
Figure 4. Schatzki ring (arrows) appears as a smooth, symmetric, ring-like constriction at the gastroesophageal junction above a hiatal hernia with a length of only 2 mm and has more abrupt borders than does a ring-like peptic stricture.
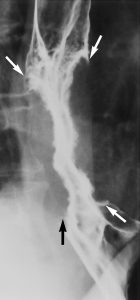
Figure 5. Infiltrating esophageal carcinoma presenting as a stricture with a markedly irregular contour and abrupt, shelflike proximal and distal margins (arrows).
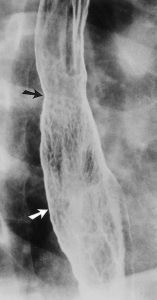
Figure 6. Barrett’s esophagus with a midesophageal stricture (black arrow) and a reticular mucosal pattern (white arrow) thought to result from intestinal metaplasia in Barrett mucosa.
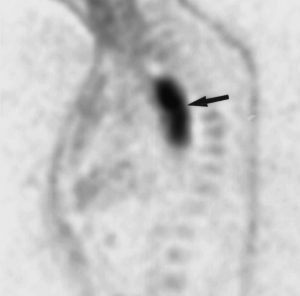
Figure 7. PET-FDG scan showing a proximal oesophageal squamous cell carcinoma.
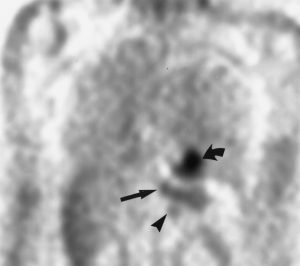
Figure 8. Coronal FDG PET showing primary adenocarcinoma of the gastroesophageal junction (straight arrow), a left-sided gastric lymph node (arrowhead) and normal uptake in the left ventricle (curved arrow).
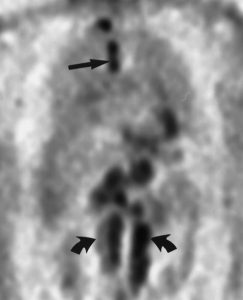
Figure 9. Coronal FDG PET shows nodal metastases in the paratracheal region (straight arrow) and extensive retroperitoneal involvement (curved arrows) from adenocarcinoma of the gastroesophageal junction.

