Updates on the management of COPD
Updates on the management of COPD – Cynthia Farrugia Jones
Introduction
Chronic Obstructive Pulmonary disease has long been defined by chronic bronchitis and emphysema, with patients being placed in one category or the other. This excluded the airflow obstruction which is predominant in COPD. Recent studies show different mechanisms of airflow obstruction: [1] loss of support of the small airways in emphysema, [2] chronic inflammation taking part in the smaller airways, and [3] presence of mucus in the small airways.1 COPD has now a more flexible definition of preventable and treatable disease with airflow limitation that is not fully reversible and an inflammatory response to noxious particles.
Updates on the management of COPD
Smoking still remains the primary culprit but with a more complicated association, since as reported in the literature, only 20-40% of those exposed to cigarette smoke develop COPD.2,3 Many patients in certain parts of the world have never smoked but have been exposed to wood flame during cooking. Thus, the etiology of COPD includes the effect of harmful agents, genetic predisposition, infectious agents and airway hyper-responsiveness.4
Currently COPD is the 6th leading cause of death, but for the year 2020, it is estimated to be ranked third.5 This is partly being attributed to the gaining popularity of water pipe smoking (Figure 1) amongst youths in the Middle East.
According to the WHO Study Group on Tobacco Product Regulation, a typical one hour-long session of water pipe smoking involves inhaling 100-200 times the volume of smoke inhaled by one cigarette.
Figure 1. A Middle-East water pipe

Lung function decline
Fletcher and Peto (Figure 2) demonstrated that smokers have a more accelerated loss in lung function, which ultimately leads to symptomatic COPD.6 The loss of lung function is thought to be about 60 ml of forced expiratory volume in 1 second (FEV1) per year, as compared to the normal loss of lung function, which is about 30 ml per year. This study6 showed that 15% of smokers are susceptible to COPD.
Figure 2. Fletcher and Peto graph of lung function decline
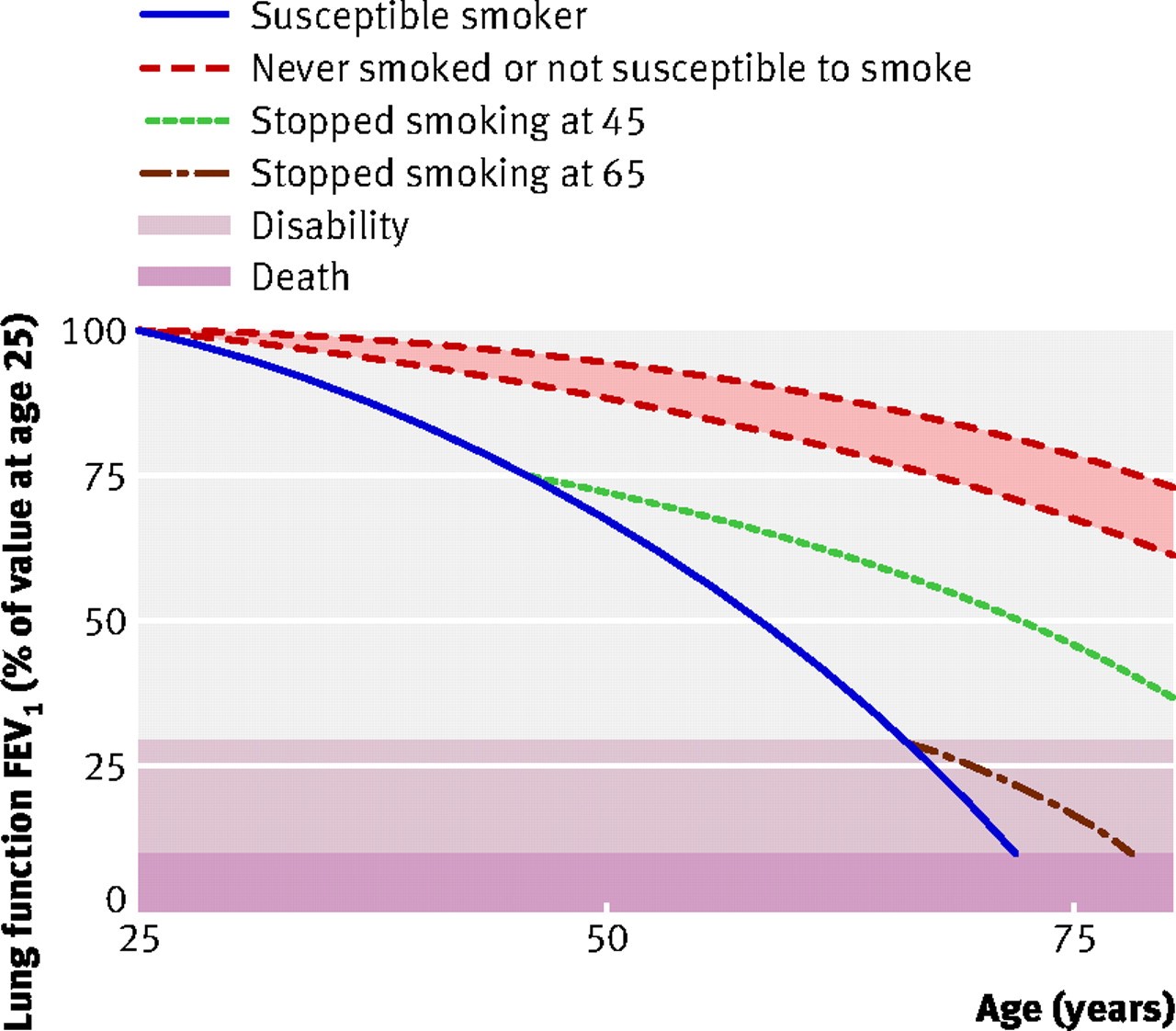
Recent studies report the proportion of smokers who are susceptible to COPD to be 30-40%. Anthonisen et al. found that people who quit smoking had a small loss of lung function of only 27 ml a year, as compared to those who continued to smoke, who lost about 60 ml per year.7 Regarding the ones who quit intermittently, the study found their loss of lung function comparable to those who never quit smoking, which means that smoking should be completely discontinued for patients to obtain the full benefit of doing so.
The most recent data is from the ECLIPSE study.8 The mean decline in lung function in smokers was actually less than expected, averaging approximately 33 ml per year, with only 38% of patients reaching a FEV1 decline of more than 40 ml per year. The reasons behind this slow decline may be related to environmental conditions or treatment.8 This data suggests that treatment probably does have a positive impact on the lung function of COPD patients. There is increasing interest in the frequency of COPD exacerbations, since this was found to relate to prevention and treatment. Most of the new drugs being investigated and developed are targeting patients with the ‘frequent-exacerbation’ phenotype.
The ECLIPSE data, showed that, depending on the overall Global Initiative for Obstructive Lung Disease (GOLD) stage, a significant proportion of patients had 2 or more exacerbations per year, and were thus defined as frequent exacerbators.9
More than 75% of treated patients in these studies were given long-acting bronchodilators and/or inhaled steroids. The method to identify the frequent exacerbators in clinical practice is their history. Other parameters that are helpful to identify these frequent exacerbators include having a more severe lung function, a worsening quality of life and a high white cell count.
An interesting finding is that having a history of gastroesophageal reflux disease (GERD) or heartburn seems to correlate with the development of COPD exacerbations.10 The pathogenesis of this is poorly understood, but may be related to a swallowing dysfunction. Although no studies have been conducted to evaluate whether treating these patients would help with COPD exacerbations, it is best to treat these patients with a trial of anti-reflux medicines.
Data from the COPD Gene study demonstrated that having chronic bronchitis doubles the frequency of COPD exacerbations.11 Therefore, patients with a chronic bronchitis phenotype have a heightened risk and should be identified and followed-up closely. The COPD Gene study studied 10,000 people with COPD and conducted gene sequencing but it was not possible to identify a specific gene. In exacerbations one needs to consider other etiologies including pneumonia, congestive heart failure exacerbation, pulmonary emboli, as well as simply, non-compliance to prescribed medicines.12
The ECLIPSE study also reported that infections were responsible for approximately 50% of exacerbations. Haemophilus influenza along with Streptococcus pneumoniae and Moraxella catarrhalis were the most predominant. Patients with more severe disease were mainly affected by Pseudomonas infections. The role of Staphylococcus aureus and other Gram-negative bacteria is not well defined in exacerbations. It is also important to understand that some bacteria are colonizers and not actual pathogens. Previously, it was thought that COPD exacerbations were driven by a change in the concentration of bacteria; however, now we know that it is probably due to acquisition of new strains of bacteria.13
Management
The aim is to relieve symptoms, improve exercise tolerance and improve the overall health status. The main goals in the treatment of COPD include reducing the risks, preventing disease progression, preventing frequent exacerbations and reducing mortality. The pharmacological options in COPD are a growing field.14
Twenty years ago, the only treatment options were short-acting β-agonists, short-acting anti-muscarinics or a combination of both, in addition to theophylline and oral steroids. Nowadays, the list includes drugs such as long-acting bronchodilators (salmeterol, formoterol and once-daily indacaterol), inhaled corticosteroids (ICS) and phosphodiesterase inhibitors.
FEV1 is no longer a comprehensive measurement of COPD disease. In a study by Westwood et al., there was a modest relationship between an increase in FEV1 and improvements in the St George’s Respiratory Questionnaire. This means that treatment effectiveness can be assessed at a study level;15 however, from a practical point of view, this may be contradicted. For example, a patient may have an FEV1 of 40%, yet their St George’s Respiratory Questionnaire score may be almost normal, compared to another patient with the same FEV1 who is disabled and terribly symptomatic.
Other tests, besides measuring the lung function, give a better idea of the disease activity and severity. The BODE index,16 specific quality-of-life indices such as symptom rating scales, and also exercise testing can all give a better assessment of the lung function. However, the limitations of these tests are that they are often difficult to perform in the clinic setting. The new GOLD guidelines recommend the Modified Medical Research Council Dyspnoea (MMRC) scale and the COPD Assessment Test (CAT) for use in the assessment of symptoms in COPD independently of FEV1.
The UPLIFT study found that tiotropium, a bronchodilator, can actually cause a significant reduction of about 14% of exacerbations.17 A study performed by Calverley et al. compared formoterol–budesonide versus formoterol alone in the reduction of symptoms, and found that the combination of a long-acting β-agonist (LABA) with ICS reduces COPD symptoms on a daily basis.18 Further to this, the FLAME study19 showed that the combination LABA with a long-acting muscarinic antagonist (LAMA) (indacaterol-glycopyrronium) showed consistent superiority to LABA-ICS (salmeterol-fluticasone) for outcomes of exacerbations, lung function and health status. Thus, it is important not to have strict categorization of these drug classes. There are also multiple new drugs that do not act on the symptoms but have a significant impact on reducing exacerbations, such as the recently introduced oral phosphodiesterase inhibitor, roflumilast.
How have the guidelines changed?
COPD is no longer measured just by using spirometry and FEV1. The full picture should take in account the assessment of symptoms and exacerbations. Associated co-morbidities, which are potential factors, also need to be included in the patients’ assessment. Subsequently, the best assessment of COPD is to combine FEV1 and symptom severity. (Figure 3 COPD groups) In summary, group A has an FEV1 that is still above 50%, exacerbations that are not frequent and they are generally not symptomatic.
Group B is the same, except that they have more symptoms. Group C has a lower FEV1, along with frequent exacerbations or symptoms, whereas group D combines all parameters.
Figure 3. COPD groups
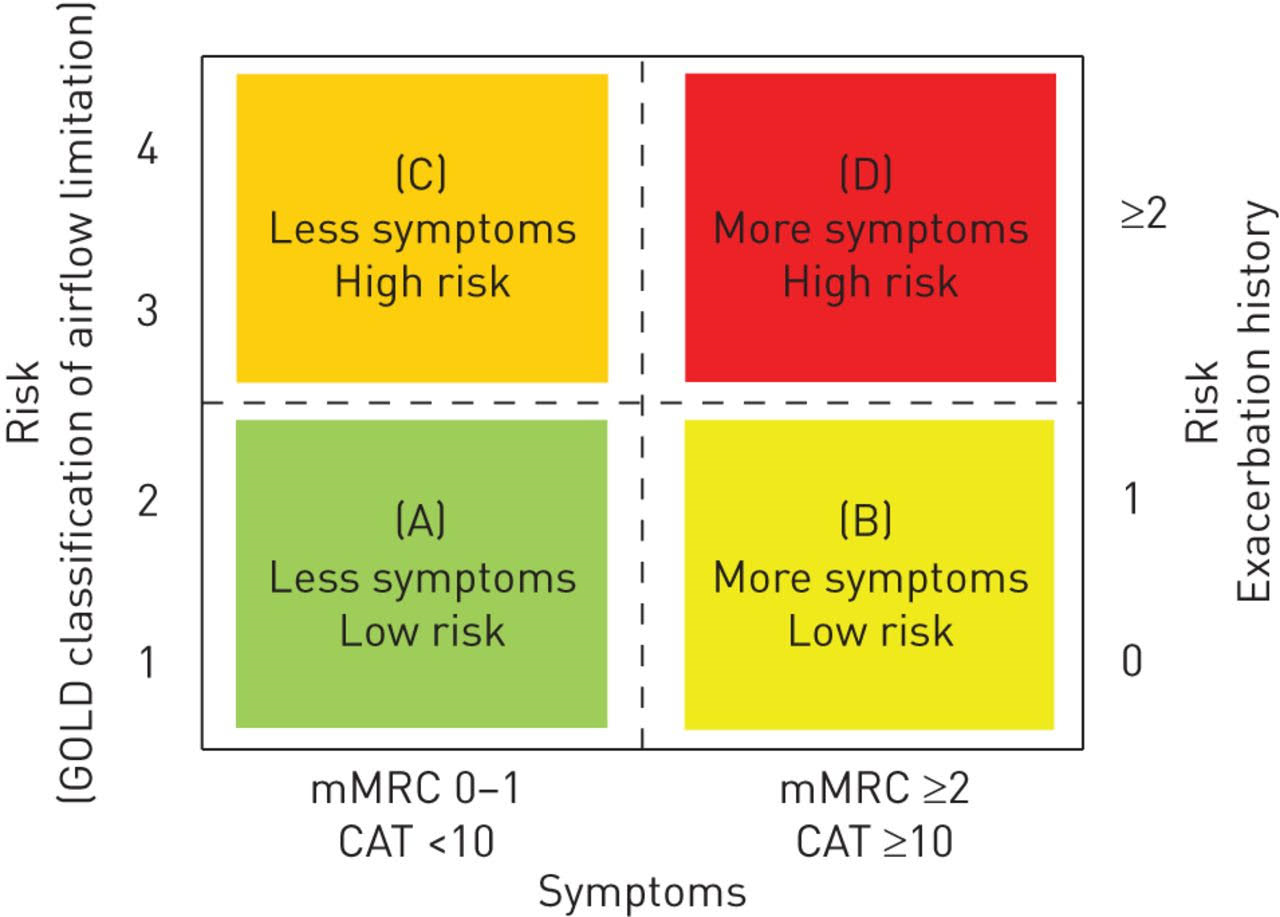
The difficulty arises in deciding on the pharmacological treatment for each group. Generally, group A patients receive short-acting bronchodilators, whereas group B must be given LAMA or a LABA, and groups C and D should take an ICS in combination.
Conclusion
The disease progression of COPD should be explained to patients to help them overcome the denial of the causality of smoking. It is very important for patients to stay active; patients with symptomatic airflow obstruction should be offered a rehabilitation program as part of an optimal treatment plan.
It is difficult to treat dyspnoea or completely eliminate it, and this can require the use of double combinations of bronchodilators, i.e. LAMA plus LABA. If the patient does not respond, then a triple combination is recommended, with the addition of ICS. Some patients still do not improve, and one should consider adding a low dose of theophylline.20,21 Sometimes, nebulized bronchodilators may help as a back-up management. The last resort remains the use of chronic oral corticosteroids; however, this is discouraged due to the side-effects.
When the problem relates to recurrent exacerbations, a combination of ICS plus LABA / phosphodiesterase inhibitor may be used, as well as the addition of prophylactic antibiotics.
References
Updates on the management of COPD
Cynthia Farrugia Jones
Introduction
Chronic Obstructive Pulmonary disease has long been defined by chronic bronchitis and emphysema, with patients being placed in one category or the other. This excluded the airflow obstruction which is predominant in COPD. Recent studies show different mechanisms of airflow obstruction: [1] loss of support of the small airways in emphysema, [2] chronic inflammation taking part in the smaller airways, and [3] presence of mucus in the small airways.1 COPD has now a more flexible definition of preventable and treatable disease with airflow limitation that is not fully reversible and an inflammatory response to noxious particles.
Updates on the management of COPD
Smoking still remains the primary culprit but with a more complicated association, since as reported in the literature, only 20-40% of those exposed to cigarette smoke develop COPD.2,3 Many patients in certain parts of the world have never smoked but have been exposed to wood flame during cooking. Thus, the etiology of COPD includes the effect of harmful agents, genetic predisposition, infectious agents and airway hyper-responsiveness.4
Currently COPD is the 6th leading cause of death, but for the year 2020, it is estimated to be ranked third.5 This is partly being attributed to the gaining popularity of water pipe smoking (Figure 1) amongst youths in the Middle East.
According to the WHO Study Group on Tobacco Product Regulation, a typical one hour-long session of water pipe smoking involves inhaling 100-200 times the volume of smoke inhaled by one cigarette.
Figure 1. A Middle-East water pipe
Lung function decline
Fletcher and Peto (Figure 2) demonstrated that smokers have a more accelerated loss in lung function, which ultimately leads to symptomatic COPD.6 The loss of lung function is thought to be about 60 ml of forced expiratory volume in 1 second (FEV1) per year, as compared to the normal loss of lung function, which is about 30 ml per year. This study6 showed that 15% of smokers are susceptible to COPD.
Figure 2. Fletcher and Peto graph of lung function decline
Recent studies report the proportion of smokers who are susceptible to COPD to be 30-40%. Anthonisen et al. found that people who quit smoking had a small loss of lung function of only 27 ml a year, as compared to those who continued to smoke, who lost about 60 ml per year.7 Regarding the ones who quit intermittently, the study found their loss of lung function comparable to those who never quit smoking, which means that smoking should be completely discontinued for patients to obtain the full benefit of doing so.
The most recent data is from the ECLIPSE study.8 The mean decline in lung function in smokers was actually less than expected, averaging approximately 33 ml per year, with only 38% of patients reaching a FEV1 decline of more than 40 ml per year. The reasons behind this slow decline may be related to environmental conditions or treatment.8 This data suggests that treatment probably does have a positive impact on the lung function of COPD patients. There is increasing interest in the frequency of COPD exacerbations, since this was found to relate to prevention and treatment. Most of the new drugs being investigated and developed are targeting patients with the ‘frequent-exacerbation’ phenotype.
The ECLIPSE data, showed that, depending on the overall Global Initiative for Obstructive Lung Disease (GOLD) stage, a significant proportion of patients had 2 or more exacerbations per year, and were thus defined as frequent exacerbators.9
More than 75% of treated patients in these studies were given long-acting bronchodilators and/or inhaled steroids. The method to identify the frequent exacerbators in clinical practice is their history. Other parameters that are helpful to identify these frequent exacerbators include having a more severe lung function, a worsening quality of life and a high white cell count.
An interesting finding is that having a history of gastroesophageal reflux disease (GERD) or heartburn seems to correlate with the development of COPD exacerbations.10 The pathogenesis of this is poorly understood, but may be related to a swallowing dysfunction. Although no studies have been conducted to evaluate whether treating these patients would help with COPD exacerbations, it is best to treat these patients with a trial of anti-reflux medicines.
Data from the COPD Gene study demonstrated that having chronic bronchitis doubles the frequency of COPD exacerbations.11 Therefore, patients with a chronic bronchitis phenotype have a heightened risk and should be identified and followed-up closely. The COPD Gene study studied 10,000 people with COPD and conducted gene sequencing but it was not possible to identify a specific gene. In exacerbations one needs to consider other etiologies including pneumonia, congestive heart failure exacerbation, pulmonary emboli, as well as simply, non-compliance to prescribed medicines.12
The ECLIPSE study also reported that infections were responsible for approximately 50% of exacerbations. Haemophilus influenza along with Streptococcus pneumoniae and Moraxella catarrhalis were the most predominant. Patients with more severe disease were mainly affected by Pseudomonas infections. The role of Staphylococcus aureus and other Gram-negative bacteria is not well defined in exacerbations. It is also important to understand that some bacteria are colonizers and not actual pathogens. Previously, it was thought that COPD exacerbations were driven by a change in the concentration of bacteria; however, now we know that it is probably due to acquisition of new strains of bacteria.13
Management
The aim is to relieve symptoms, improve exercise tolerance and improve the overall health status. The main goals in the treatment of COPD include reducing the risks, preventing disease progression, preventing frequent exacerbations and reducing mortality. The pharmacological options in COPD are a growing field.14
Twenty years ago, the only treatment options were short-acting β-agonists, short-acting anti-muscarinics or a combination of both, in addition to theophylline and oral steroids. Nowadays, the list includes drugs such as long-acting bronchodilators (salmeterol, formoterol and once-daily indacaterol), inhaled corticosteroids (ICS) and phosphodiesterase inhibitors.
FEV1 is no longer a comprehensive measurement of COPD disease. In a study by Westwood et al., there was a modest relationship between an increase in FEV1 and improvements in the St George’s Respiratory Questionnaire. This means that treatment effectiveness can be assessed at a study level;15 however, from a practical point of view, this may be contradicted. For example, a patient may have an FEV1 of 40%, yet their St George’s Respiratory Questionnaire score may be almost normal, compared to another patient with the same FEV1 who is disabled and terribly symptomatic.
Other tests, besides measuring the lung function, give a better idea of the disease activity and severity. The BODE index,16 specific quality-of-life indices such as symptom rating scales, and also exercise testing can all give a better assessment of the lung function. However, the limitations of these tests are that they are often difficult to perform in the clinic setting. The new GOLD guidelines recommend the Modified Medical Research Council Dyspnoea (MMRC) scale and the COPD Assessment Test (CAT) for use in the assessment of symptoms in COPD independently of FEV1.
The UPLIFT study found that tiotropium, a bronchodilator, can actually cause a significant reduction of about 14% of exacerbations.17 A study performed by Calverley et al. compared formoterol–budesonide versus formoterol alone in the reduction of symptoms, and found that the combination of a long-acting β-agonist (LABA) with ICS reduces COPD symptoms on a daily basis.18 Further to this, the FLAME study19 showed that the combination LABA with a long-acting muscarinic antagonist (LAMA) (indacaterol-glycopyrronium) showed consistent superiority to LABA-ICS (salmeterol-fluticasone) for outcomes of exacerbations, lung function and health status. Thus, it is important not to have strict categorization of these drug classes. There are also multiple new drugs that do not act on the symptoms but have a significant impact on reducing exacerbations, such as the recently introduced oral phosphodiesterase inhibitor, roflumilast.
How have the guidelines changed?
COPD is no longer measured just by using spirometry and FEV1. The full picture should take in account the assessment of symptoms and exacerbations. Associated co-morbidities, which are potential factors, also need to be included in the patients’ assessment. Subsequently, the best assessment of COPD is to combine FEV1 and symptom severity. (Figure 3 COPD groups) In summary, group A has an FEV1 that is still above 50%, exacerbations that are not frequent and they are generally not symptomatic.
Group B is the same, except that they have more symptoms. Group C has a lower FEV1, along with frequent exacerbations or symptoms, whereas group D combines all parameters.
Figure 3. COPD groups
The difficulty arises in deciding on the pharmacological treatment for each group. Generally, group A patients receive short-acting bronchodilators, whereas group B must be given LAMA or a LABA, and groups C and D should take an ICS in combination.
Conclusion
The disease progression of COPD should be explained to patients to help them overcome the denial of the causality of smoking. It is very important for patients to stay active; patients with symptomatic airflow obstruction should be offered a rehabilitation program as part of an optimal treatment plan.
It is difficult to treat dyspnoea or completely eliminate it, and this can require the use of double combinations of bronchodilators, i.e. LAMA plus LABA. If the patient does not respond, then a triple combination is recommended, with the addition of ICS. Some patients still do not improve, and one should consider adding a low dose of theophylline.20,21 Sometimes, nebulized bronchodilators may help as a back-up management. The last resort remains the use of chronic oral corticosteroids; however, this is discouraged due to the side-effects.
When the problem relates to recurrent exacerbations, a combination of ICS plus LABA / phosphodiesterase inhibitor may be used, as well as the addition of prophylactic antibiotics.
References
1.Szilasi M, Dolinay T, Nemes Z, Strausz J. Pathology of chronic obstructive pulmonary disease. Pathol. Oncol. Res. 2006; 12(1):52-60.
- Lokke A, Lange P, Scharling H, Fabricius P, Vestbo J. Developing COPD: a 25 year follow up study of the general population. Thorax 2006; 61(11):935-939.
- Pauwels RA, Rabe KF, Burden and clinical features of chronic obstructive pulmonary disease (COPD). Lancet 2004; 364(9434):613-620.
- Huertas A, Palange P. COPD: a multifactorial systemic disease. Ther. Adv. Respir. 2011; 5(3):217224.
- Gershon AS, Warner L, Cascagnette P, Victor JC, To T. Lifetime risk of developing chronic obstructive pulmonary disease: a longitudinal population study.Lancet2011; 378(9795):991–996.
- Fletcher C, Peto R. The natural history of chronic airflow obstruction.Br. Med. J. 1977; 1(6077):1645–1648.
- Anthonisen NR, Connett JE, Murray RP. Smoking and lung function of Lung Health Study participants after 11 years.Am. J. Respir. Crit. Care Med. 2002; 166(5):675–679.
8.Vestbo J, Edwards LD, Scanlon PD et al. Changes in forced expiratory volume in 1 second over time in COPD.N. Engl. J. Med. 2011; 365(13):1184–1192.
9.Hurst JR, Vestbo J, Anzueto A et al. Susceptibility to exacerbation in chronic obstructive pulmonary disease. N. Engl. J. Med. 2010; 363(12):1128–1138.
- Liang B, Wang M, Yi Q, Feng Y. Association of gastroesophageal reflux disease risk with exacerbations of chronic obstructive pulmonary disease.Dis. Esophagus 2013; 26(6):557-60.
- Kim V, Han MK, Vance GBet al. The chronic bronchitic phenotype of COPD: an analysis of the COPDGene Study.Chest 2011; 140(3):626–633.
- Sethi S, Evans N, Grant BJ, Murphy TF. New strains of bacteria and exacerbations of chronic obstructive pulmonary disease.N. Engl. J. Med. 2002; 347(7):465–471.
- Sethi S, Murphy TF. Infection in the pathogenesis and course of chronic obstructive pulmonary disease.N. Engl. J. Med. 2008; 359(22):2355–2365.
- Hanania NA, Donohue JF. Pharmacologic interventions in chronic obstructive pulmonary disease: bronchodilators.Proc. Am. Thorac. Soc. 2007; 4(7):526–534.
- Westwood M, Bourbeau J, Jones Pw et al.Relationship between FEV1 change and patient-reported outcomes in randomised trials of inhaled bronchodilators for stable COPD: a systematic review.Respir. Res. 2011; 12:40.
- Cote CG, Celli BR. BODE index: a new tool to stage and monitor progression of chronic obstructive pulmonary disease.Pneumonol. Alergol. Pol. 2009; 77(3):305–313.
- Jones PW, Harding G, Berry P et al. Development and first validation of the COPD Assessment Test.Eur. Respir. J. 2009; 34(3):648–654.
- Calverley PM, Boonsawat W, Cseke Z et al. Maintenance therapy with budesonide and formoterol in chronic obstructive pulmonary disease.Eur. Respir. J. 2003; 22(6):912–919.
- Wedzicha JA, Banerji D, Chapman KR, Vestbo J, Roche N, Ayers RT, Thach C, Fogel R, Patalano F, Vogelmeier CF; FLAME Investigators. Indacaterol-Glycopyrronium versus Salmeterol-Fluticasone for COPD. N Engl J Med 2016;374(23):2222-34.
- Cosio BG, Iglesias A, Rios Aet al. Low-dose theophylline enhances the anti-inflammatory effects of steroids during exacerbations of COPD.Thorax 2009; 64(5):424–429.
- Kanehara M, Yokoyama A, Tomoda Yet al. Anti-inflammatory effects and clinical efficacy of theophylline and tulobuterol in mild-to-moderate chronic obstructive pulmonary disease.Pulm. Pharmacol. Ther. 2008; 21(6):874–878.
1.Szilasi M, Dolinay T, Nemes Z, Strausz J. Pathology of chronic obstructive pulmonary disease. Pathol. Oncol. Res. 2006; 12(1):52-60.
- Lokke A, Lange P, Scharling H, Fabricius P, Vestbo J. Developing COPD: a 25 year follow up study of the general population. Thorax 2006; 61(11):935-939.
- Pauwels RA, Rabe KF, Burden and clinical features of chronic obstructive pulmonary disease (COPD). Lancet 2004; 364(9434):613-620.
- Huertas A, Palange P. COPD: a multifactorial systemic disease. Ther. Adv. Respir. 2011; 5(3):217224.
- Gershon AS, Warner L, Cascagnette P, Victor JC, To T. Lifetime risk of developing chronic obstructive pulmonary disease: a longitudinal population study.Lancet2011; 378(9795):991–996.
- Fletcher C, Peto R. The natural history of chronic airflow obstruction.Br. Med. J. 1977; 1(6077):1645–1648.
- Anthonisen NR, Connett JE, Murray RP. Smoking and lung function of Lung Health Study participants after 11 years.Am. J. Respir. Crit. Care Med. 2002; 166(5):675–679.
8.Vestbo J, Edwards LD, Scanlon PD et al. Changes in forced expiratory volume in 1 second over time in COPD.N. Engl. J. Med. 2011; 365(13):1184–1192.
9.Hurst JR, Vestbo J, Anzueto A et al. Susceptibility to exacerbation in chronic obstructive pulmonary disease. N. Engl. J. Med. 2010; 363(12):1128–1138.
- Liang B, Wang M, Yi Q, Feng Y. Association of gastroesophageal reflux disease risk with exacerbations of chronic obstructive pulmonary disease.Dis. Esophagus 2013; 26(6):557-60.
- Kim V, Han MK, Vance GBet al. The chronic bronchitic phenotype of COPD: an analysis of the COPDGene Study.Chest 2011; 140(3):626–633.
- Sethi S, Evans N, Grant BJ, Murphy TF. New strains of bacteria and exacerbations of chronic obstructive pulmonary disease.N. Engl. J. Med. 2002; 347(7):465–471.
- Sethi S, Murphy TF. Infection in the pathogenesis and course of chronic obstructive pulmonary disease.N. Engl. J. Med. 2008; 359(22):2355–2365.
- Hanania NA, Donohue JF. Pharmacologic interventions in chronic obstructive pulmonary disease: bronchodilators.Proc. Am. Thorac. Soc. 2007; 4(7):526–534.
- Westwood M, Bourbeau J, Jones Pw et al.Relationship between FEV1 change and patient-reported outcomes in randomised trials of inhaled bronchodilators for stable COPD: a systematic review.Respir. Res. 2011; 12:40.
- Cote CG, Celli BR. BODE index: a new tool to stage and monitor progression of chronic obstructive pulmonary disease.Pneumonol. Alergol. Pol. 2009; 77(3):305–313.
- Jones PW, Harding G, Berry P et al. Development and first validation of the COPD Assessment Test.Eur. Respir. J. 2009; 34(3):648–654.
- Calverley PM, Boonsawat W, Cseke Z et al. Maintenance therapy with budesonide and formoterol in chronic obstructive pulmonary disease.Eur. Respir. J. 2003; 22(6):912–919.
- Wedzicha JA, Banerji D, Chapman KR, Vestbo J, Roche N, Ayers RT, Thach C, Fogel R, Patalano F, Vogelmeier CF; FLAME Investigators. Indacaterol-Glycopyrronium versus Salmeterol-Fluticasone for COPD. N Engl J Med 2016;374(23):2222-34.
- Cosio BG, Iglesias A, Rios Aet al. Low-dose theophylline enhances the anti-inflammatory effects of steroids during exacerbations of COPD.Thorax 2009; 64(5):424–429.
- Kanehara M, Yokoyama A, Tomoda Yet al. Anti-inflammatory effects and clinical efficacy of theophylline and tulobuterol in mild-to-moderate chronic obstructive pulmonary disease.Pulm. Pharmacol. Ther. 2008; 21(6):874–878.

