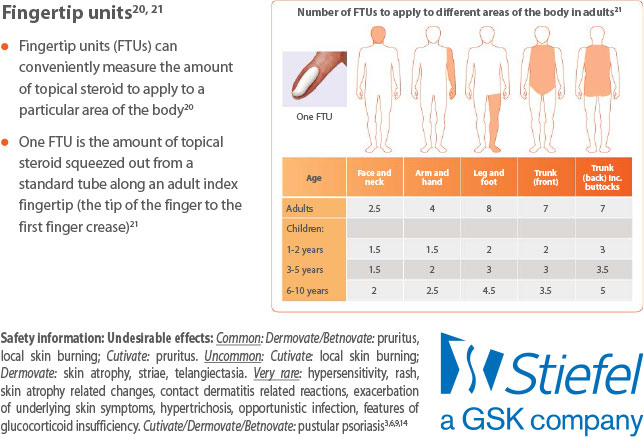
GSK topical steroid preparations

 |
|||
|
Dear Healthcare Professional,
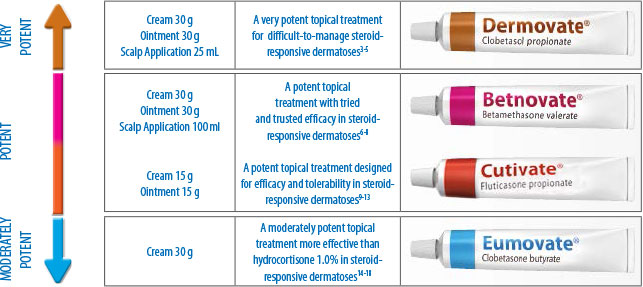
As you probably know, itchiness and skin discomfort associated with dermatoses can interfere with the daily activities of patients.1,2The Stiefel family of topical steroids, with a range of potencies to suit the varying needs of your patients, helps control their symptoms.3–6
The table below briefly highlights the products available within the Stiefel family of topical steroids:
|
|||
|
|
|||
|
Our range of potencies allows a stepwise approach to clinical management of patients when required.19To find out more about our family of topical steroids, please refer to the full summary of product characteristics (SPCs) (link below).
|
|||
|
|
|||
|
|||
|
|||
| Job No: MLT_GIB/FPC/0001/15 Date of Preparation: December 2015 |
|||
|
|