Companies Plan To Resume Renal Denervation Trials
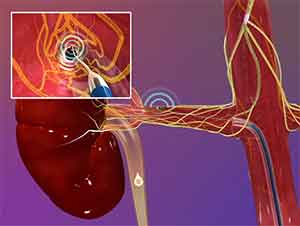 Medtronic and Boston Scientific have announced plans to start phase II clinical trials this year for their updated renal denervation catheters. The once promising new technology is intended to treat hypertension unresponsive to drug therapy. The failure last year of Medtronic’s SYMPLICITY HTN-3, the first large pivotal trial to rigorously test renal denervation, sent manufacturers back to their drawing boards to redesign the devices.
Medtronic and Boston Scientific have announced plans to start phase II clinical trials this year for their updated renal denervation catheters. The once promising new technology is intended to treat hypertension unresponsive to drug therapy. The failure last year of Medtronic’s SYMPLICITY HTN-3, the first large pivotal trial to rigorously test renal denervation, sent manufacturers back to their drawing boards to redesign the devices.
Last week, Medtronic disclosed in its quarterly report that it planned to submit to the FDA its application for an investigational device exemption for its next-generation renal denervation device. The spiral-shaped device has multiple electrodes and is designed to achieve more comprehensive ablation of the renal arteries. The company is seeking FDA approval to proceed with a phase II trial.
In its quarterly report, Boston Scientific said it plans to begin enrolling patients in the first half of this year in a phase II trial, REDUCE-HTN REINFORCE, testing its Vessix platform.
Background: NEJM Journal Watch General Medicine summary on SYMPLICITY HTN-3

